Licensed Stem Cell Clinic Mexico: A Patient Safety Guide
You are probably reading this after comparing clinics, scanning websites, and trying to answer one hard question. Which licensed stem cell clinic Mexico patients can trust?
That question matters more than many realize. Mexico offers access to advanced regenerative care, especially for patients from the United States and Canada seeking options for chronic pain, orthopedic injuries, autoimmune concerns, recovery, and longevity support. The opportunity is real. The risk is real too.
The market is growing quickly. The Mexico stem cells market generated USD 160.0 million in revenue in 2023 and is projected to reach USD 369.8 million by 2030, reflecting a CAGR of 12.7%, driven by Mexico’s role as a medical tourism destination for advanced stem cell treatments for patients from the US and Canada, according to Grand View Research’s Mexico stem cells market outlook.
Growth attracts serious clinics. It also attracts aggressive marketing.
Patients often contact our field after seeing polished promises online. A website may look advanced. The clinic may mention doctors, laboratory standards, and “advanced protocols.” But presentation is not proof. In regenerative medicine, licensing, lab quality, physician oversight, and transparent documentation are what separate a well-run medical program from a risky purchase.
This guide is written from that practical clinical perspective. Not to create fear, but to replace confusion with a reliable screening framework. If you are considering stem cell therapy in Mexico, your safest path is to verify the clinic the way a careful medical team would verify a vendor, a lab partner, or a surgical facility.
The Promise and Peril of Stem Cell Therapy in Mexico
A patient with knee arthritis, shoulder degeneration, lupus, or chronic inflammatory symptoms often arrives at the same crossroads. Their symptoms have gone on too long. Conventional care has helped only partially. They are not looking for hype. They are looking for a clinic that handles regenerative medicine like medicine.
Mexico can be an excellent option for that patient. It is one of the places where advanced allogeneic stem cell programs are accessible in a more practical way than many patients find at home. For the right person, that can mean meaningful access to physician-guided care, imaging, laboratory support, and a more extensive recovery plan.
The problem is not Mexico itself. The problem is that many patients shop by headline claims instead of medical structure.
A glossy clinic page may highlight stem cells for osteoarthritis, sports recovery, autoimmune support, or longevity optimization. That does not tell you whether the facility is operating under the right regulatory permissions, whether the cells were processed correctly, or whether a physician is directing the treatment plan. Those details affect safety long before the first injection or infusion.
What works
In practice, the clinics that inspire confidence tend to share a few traits:
Clear licensure: They explain what they are licensed to do.
Defined medical leadership: Physicians, not sales teams, drive the clinical plan.
Transparent cell handling: They can describe sourcing, processing, and documentation for allogeneic products.
Real diagnostics: Imaging, labs, and follow-up are part of care, not optional extras.
What does not work
Other patterns should make you pause:
Condition lists without medical detail
Evasive answers about lab standards
One-size-fits-all protocols
Promises that sound absolute
A strong regenerative program does not begin with a promise. It begins with verification.
That is the lens to use throughout your search for a licensed stem cell clinic Mexico patients can trust with confidence.
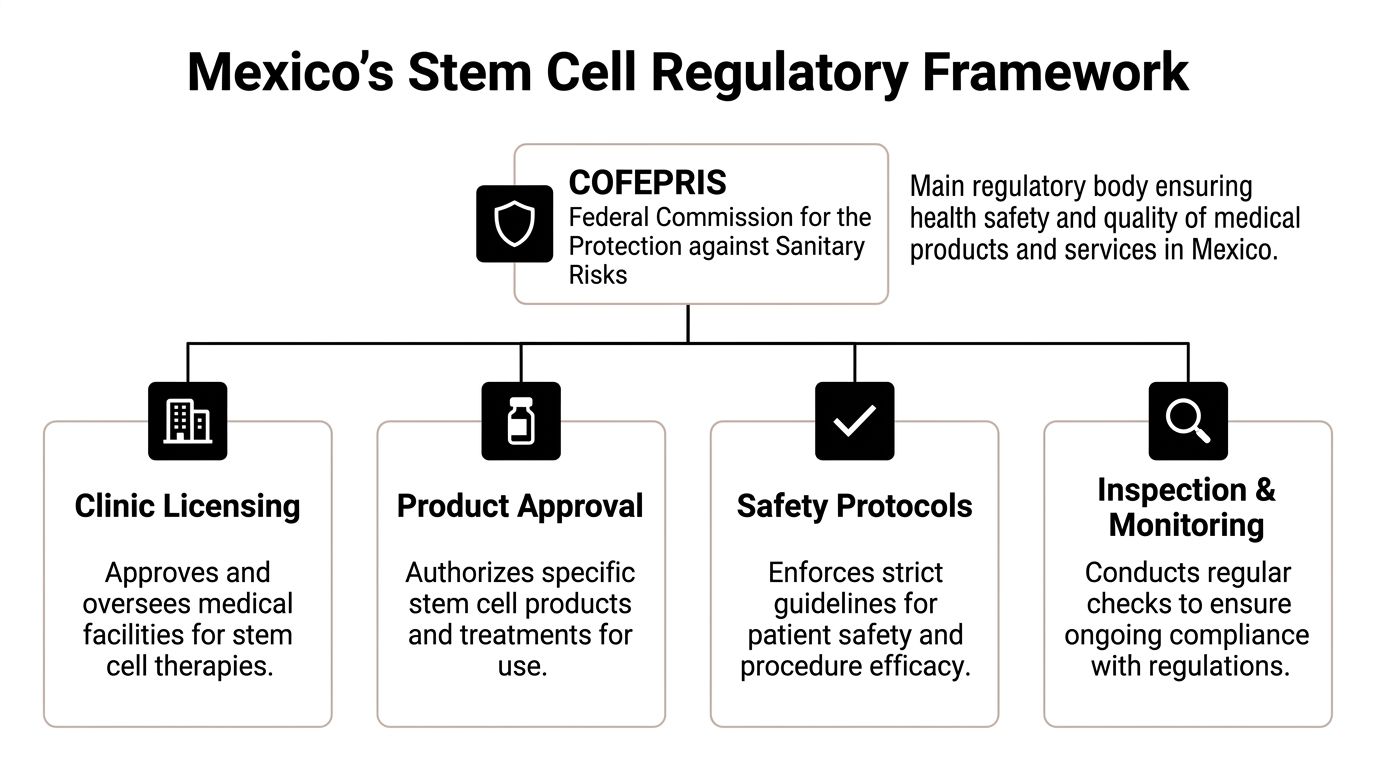
Understanding Mexico's Stem Cell Regulatory Framework
The central regulator patients need to know is COFEPRIS, the Federal Commission for the Protection against Sanitary Risks. In practical terms, it functions as Mexico’s key health authority for oversight of medical products, facilities, and safety standards.
If you are evaluating a stem cell clinic, COFEPRIS is not a minor administrative detail. It is the first checkpoint.
Why patients cannot rely on website claims alone
A real-world example shows why independent verification matters. A 2021 study of 76 stem cell clinics in Tijuana found that 13 claimed COFEPRIS licensing on their websites, only 6 matched the official government registry by name or address, and only 1 matched both precisely, according to this peer-reviewed analysis of stem cell clinic marketing and licensing claims in Tijuana.
That gap is not academic. It is exactly why patients should never stop at “licensed” language on a clinic website.
A clinic may use regulatory terms loosely. It may refer to approvals that do not cover the service being sold. It may mention a lab relationship without explaining whether the treatment facility itself holds the proper authorizations. Those distinctions matter.
What COFEPRIS oversight means in practical terms
For patients, COFEPRIS oversight should be understood as a framework that touches several parts of care:
Facility oversight: The medical setting itself must operate within approved standards.
Product handling: Cell-based materials require controlled processes and documentation.
Safety procedures: Sterility, handling, and adverse event readiness matter.
Inspection and compliance: A legitimate clinic should be prepared for scrutiny, not just marketing.
If a clinic cannot clearly explain how its operations fit within this framework, that is not a small issue. It suggests the patient is being asked to trust branding over evidence.
How to verify without becoming a regulator yourself
You do not need to become an expert in Mexican health law. You do need to ask specific questions.
Request the clinic’s COFEPRIS information in writing. Ask what the license covers. Ask whether the medical facility and the laboratory functions are both authorized. Ask who oversees the cell processing and who performs the procedure. If a clinic has a dedicated laboratory, patients should also understand how that lab fits into the treatment process, which is one reason detailed pages such as this overview of a biotechnology stem cell lab in Mexico can be useful for comparison.
If a clinic wants your payment before it is willing to clarify its regulatory status, walk away.
The safest patient is not the one who trusts the most. It is the one who verifies the most.
Decoding the Credentials A Legitimate Clinic Must Have
Not all licenses mean the same thing. That is where many patients get misled.
A clinic may talk about being authorized or compliant, but a serious regenerative medicine operation usually requires multiple distinct permissions, not one generic credential. According to Parents Guide to Cord Blood’s explanation of regenerative medicine clinic licensing in Mexico, licensed stem cell clinics must secure separate COFEPRIS licenses, including a Stem Cell Bank License for processing and storage and a Regenerative Medicine License authorizing treatment administration. Those licenses verify protocols for equipment, staff training, and sterile handling, which directly reduce contamination and treatment complication risk.
The Stem Cell Bank License
This license relates to cell processing and storage.
That means the clinic, or the partner facility handling the product, is operating under standards relevant to receiving, processing, preserving, and managing stem cell materials. For patients, this is not abstract. Cell handling quality begins before your treatment day.
If a clinic cannot explain where the allogeneic cells are processed, stored, and documented, that is a problem. A premium-looking procedure room does not compensate for weak back-end controls.
The Regenerative Medicine License
This license covers the administration of regenerative therapies.
It speaks to the treatment side of the operation. That includes whether the facility is equipped appropriately, whether staff are trained, and whether systems exist for sterile technique and complication management. A clinic might have access to cells, but that does not automatically mean it is authorized to administer them in a proper regenerative care setting.
Why partial licensing is not enough
Some clinics operate in fragments. One site consults the patient. Another source provides the product. A different team performs the intervention. When that structure is poorly explained, accountability becomes blurred.
Patients should ask direct questions such as:
Who holds the processing authorization
Who holds the treatment authorization
Where are the cells stored before use
Who is responsible if there is a complication
A more integrated setup usually gives the patient a cleaner chain of custody and clearer responsibility. That matters in medicine.
What a legitimate answer sounds like
A legitimate clinic should be able to explain its credentials in plain language, without defensiveness or vague sales phrasing. It should also be able to describe how those credentials affect your care experience, from intake through procedure day and follow-up.
When patients compare providers, I advise them to read beyond surface branding and review how the clinic presents its medical model. A page such as this resource on a stem cell clinic in Mexico can help patients understand what a more structured clinical presentation looks like.
What works is clarity. What does not work is “we have approvals” without defining what those approvals authorize.
Evaluating the Science Behind the Services Lab Quality and Cell Viability
Licensure is the floor. It is not the ceiling.
Once a clinic clears the regulatory threshold, the next question is scientific quality. In stem cell medicine, much of that quality lives inside the lab. Patients usually focus on the injection, the infusion, or the procedure itself. Clinicians know the outcome starts much earlier, with how the cells were handled, protected, and documented.
According to Stem Cell Mexico’s overview of in-house ISO-certified lab standards and cell viability, top-tier clinics operate in-house, ISO-certified labs using stringent cleanroom protocols to process allogeneic mesenchymal stem cells, with post-thaw viability rates of 94-99% verified by a Certificate of Analysis. That matters because high-viability MSCs are tied to effective immunomodulation and tissue repair in orthopedic and autoimmune applications.
Why ISO standards matter to patients
Patients are not expected to memorize cleanroom terminology, but they should understand what the labels imply.
ISO-certified laboratory environments are designed to control contamination risk. In regenerative medicine, that translates into cleaner processing conditions, stronger consistency, and better quality control during handling of cellular products.
When a clinic mentions ISO 5, ISO 6, or ISO 7 environments, the practical message is this: the clinic is telling you it takes the processing environment seriously. That does not guarantee the right treatment plan for your condition, but it does tell you something important about operational discipline.
Why viability is not a marketing detail
Cell viability refers to how many cells remain alive and functional after processing and thawing.
That number matters because dead or poorly preserved cells do not support treatment quality the way properly handled cells can. A clinic that values scientific rigor should be able to provide a Certificate of Analysis or explain what quality documents accompany the product being used.
Patients should ask:
Are the cells allogeneic MSCs
Is a Certificate of Analysis available
How is viability verified
Is processing done in-house or outsourced
For those comparing providers, a look at an in-house clinical lab can help make these otherwise hidden standards more concrete.
A closer visual explanation can also help patients understand how laboratory processes support treatment quality.
What strong clinics do differently
The strongest programs do not isolate the lab from the clinic. They connect the laboratory, physician assessment, imaging, and treatment planning into one system.
That allows the medical team to answer a more useful question than “Do you offer stem cells?” The better question is “How do you determine which patient, which tissue target, which route of administration, and which supporting therapies fit the case?”
In regenerative medicine, the quality of the cells and the quality of the decision-making must rise together.
That is where many clinics fall short. They focus on access to cells but not on the medical discipline needed to use them well.
Your Patient Checklist for Choosing a Stem Cell Clinic
Patients do better when they evaluate a clinic with a written checklist instead of a general impression. This is the simplest way to compare one provider against another without getting pulled into branding.
Use the table below when screening any licensed stem cell clinic Mexico has to offer.
Clinic verification checklist
| Verification Point | What to Look For | Why It Matters |
|---|---|---|
| COFEPRIS licensure | Written confirmation of the clinic’s applicable licenses and what those licenses cover | Confirms the clinic is not relying on vague marketing language |
| Stem cell processing authority | Clear explanation of where allogeneic cells are processed and stored | Protects chain of custody and product handling quality |
| Treatment authorization | Confirmation that the facility is authorized to administer regenerative therapies | Distinguishes consultation centers from actual treatment facilities |
| ISO lab standards | Evidence of an ISO-certified or similarly controlled lab environment | Suggests stronger contamination control and operational discipline |
| Certificate of Analysis | Product documentation showing identity, viability, and quality testing | Helps the patient verify what is being used |
| Physician involvement | A real consultation with a qualified physician, not only a coordinator or salesperson | Clinical judgment should shape the protocol |
| Diagnostics | Imaging, laboratory review, and physical assessment before treatment | Good regenerative care should be personalized |
| Treatment plan clarity | A condition-specific rationale for route, target, and expected goals | Reduces one-size-fits-all medicine |
| Follow-up structure | A defined schedule for reassessment and response tracking | Recovery requires monitoring, not just a procedure day |
| Communication style | Direct, specific answers to hard questions | Transparent teams are easier to trust |
How to use the checklist well
Do not ask every question in a single rushed call. Send a short email first. Then schedule a consultation and compare the answers you receive.
Some patients also benefit from reviewing how higher-standard clinics present themselves publicly. For example, this overview of the best stem cell clinic in Mexico can help you see how clinics frame safety, diagnostics, and physician oversight when they are willing to be specific.
A simple rule for decision-making
If a clinic answers clearly, documents what it claims, and explains how it personalizes care, keep evaluating.
If it avoids specifics, changes the subject, or turns every question into a sales pitch, stop there.
Red Flags Common Warning Signs of a Risky Clinic
Patients rarely regret asking more questions. They do regret ignoring obvious warning signs.
Risky clinics usually reveal themselves early. Not always through one dramatic failure, but through a pattern of vagueness, pressure, and shortcuts. If you are trying to identify a trustworthy licensed stem cell clinic Mexico patients can safely consider, these red flags matter.
Sales pressure before medical review
A clinic should want to understand your case before recommending treatment.
If the first conversation is focused on deposits, package pricing, or urgency rather than diagnosis, imaging, symptoms, and goals, that is a bad sign. Medical decisions should not feel like travel upsells.
Vague answers about licenses or lab operations
A serious clinic can explain what it is licensed to do and how its cells are handled.
If the staff avoid specifics, speak in broad claims, or say details are “internal,” assume the transparency is not there. The same concern applies if no one can clearly explain who processes the cells or where the product documentation comes from.
Cure language
Be cautious with absolutes.
Stem cell therapy can support repair, modulation, and recovery in selected cases. It is not honest medicine to guarantee that one protocol will cure every arthritic joint, autoimmune condition, neurologic issue, or inflammatory syndrome. Overpromising often signals weak clinical discipline.
Minimal physician presence
If you never meet the doctor before treatment, pause.
A coordinator can help with logistics. A coordinator should not replace a physician-led assessment. That distinction becomes even more important in cases involving autoimmune disease, complex orthopedic injury, or mixed symptom patterns.
Pricing that feels disconnected from the medical process
Low prices can be tempting, especially for medical tourists. But regenerative medicine has real infrastructure behind it. Proper processing, sterile handling, diagnostics, physician oversight, and follow-up all require resources.
A price that seems too easy may reflect corners being cut somewhere you cannot see.
If a clinic cannot explain its safety model clearly, the patient should not assume one exists.
Patients comparing options often benefit from reviewing a broader discussion of whether stem cell therapy is safe, especially before committing to travel or treatment.
The safest decision is not always the most expensive one. It is the one supported by the clearest evidence of structure, accountability, and medical judgment.
How Longevity Medical Institute Embodies These Standards
The framework above is not theoretical. It describes what patients should look for in a real clinical environment.
At Longevity Medical Institute, that means regenerative care is built around physician oversight, documented safety processes, in-house diagnostics, and allogeneic cell therapy handled within a controlled medical system. The clinic model reflects the same standards patients should ask of any provider: licensure, laboratory discipline, procedural precision, and transparent care planning.
What that looks like in practice
The medical approach is not built around a generic stem cell menu. It begins with a patient workup.
That may include advanced imaging, laboratory review, physical medicine assessment, and physician consultation to identify whether the case is primarily orthopedic, inflammatory, autoimmune, neurologic, recovery-focused, or a combination. That matters because a knee with cartilage wear, a shoulder with tendon pathology, and a patient with broader immune dysregulation do not call for the same plan.
Why integration matters
An integrated clinic structure changes the patient experience in useful ways.
Diagnostics are closer to treatment planning: Imaging and clinical findings can guide more precise decisions.
The laboratory function is not detached from the medical team: Communication is clearer.
Follow-up has context: The same team that assessed the patient can reassess progress.
For patients, that often feels calmer and more coherent. For clinicians, it improves accountability.
Where premium care should be visible
Patients should expect premium care to show up in substance, not decor.
That means clear documentation, informed consent, physician access, carefully explained allogeneic protocols, and a realistic discussion of what stem cell therapy may and may not do for a given condition. It also means treatment plans that can combine regenerative procedures with supportive services such as ultrasound-guided interventions, rehabilitation strategies, hyperbaric support, or broader recovery programming when clinically appropriate.
A clinic does not become trustworthy because it looks refined. It becomes trustworthy when the systems behind the experience are medically sound.
The strongest regenerative clinics make safety visible, science understandable, and the patient pathway easy to follow.
That is the standard patients should carry into every consultation, whether they are considering care for osteoarthritis, tendon injury, autoimmune symptoms, neuroinflammatory concerns, or performance recovery.
Frequently Asked Questions About Stem Cell Therapy in Mexico
How do I know whether a clinic is legitimate?
Start with documentation, not testimonials.
Ask what licenses the clinic holds, what those licenses cover, whether treatment is physician-led, and whether the lab standards and product documentation can be explained clearly. A legitimate clinic should welcome those questions.
Should I expect a personalized consultation before treatment?
Yes.
A real regenerative consultation should review your diagnosis, symptom history, prior treatments, imaging when available, relevant labs, and your goals. If a clinic recommends a protocol before understanding your case, it is acting more like a seller than a medical practice.
Are all stem cell protocols the same?
No.
Even when two clinics both offer allogeneic stem cell therapy, they may differ substantially in patient selection, diagnostics, route of administration, procedural technique, follow-up, and laboratory quality. Those differences can matter as much as the treatment label itself.
What should I expect as a medical traveler?
Expect to plan more than the procedure day.
Good clinics help patients understand pre-arrival records, consultation timing, treatment day flow, recovery expectations, and follow-up communication after they return home. Travel medicine works best when logistics support the clinical plan instead of disrupting it.
How important is follow-up?
It is very important.
Regenerative medicine should not end when you leave the clinic. Follow-up helps monitor response, adjust supportive care, and document progress. It also creates a more honest record of what the treatment did for your condition.
Do clinics in Mexico publish strong long-term outcome data?
This remains one of the industry’s biggest weak points. According to R3 Stem Cell’s discussion of stem cell therapy in Mexico and the limits of long-term outcome reporting, a significant gap in the field is the lack of peer-reviewed, long-term efficacy data from most clinics, and very few publish condition-specific outcomes at 1, 2, or 5-year intervals. That is why patients should place extra value on clinics that track outcomes and discuss durability openly instead of relying only on procedure counts.
Does a higher-end clinic automatically mean better care?
Not automatically.
Premium presentation can improve comfort, privacy, and coordination. However, value comes from what supports the patient medically: licensed operations, quality-controlled labs, physician leadership, advanced diagnostics, and structured follow-up.
What is the most practical final question to ask a clinic?
Ask this: “Can you walk me through exactly how you determine whether I am an appropriate candidate, how the cells are documented, and how you will follow my case after treatment?”
That question often tells you more than a long brochure.
If you want help evaluating whether stem cell therapy in Mexico is appropriate for your condition, Longevity Medical Institute offers physician-led consultations, advanced diagnostics, and guidance on how a properly structured regenerative program should be assessed before treatment.
Author
Dr. Kirk Sanford, DC, Founder & CEO, Longevity Medical Institute. Dr. Sanford focuses on patient education in regenerative and longevity medicine, translating complex therapies into clear, practical guidance for patients.
Medical Review
Dr. Félix Porras, MD, Medical Director, Longevity Medical Institute. Dr. Porras provides clinical oversight and medical review to help ensure accuracy, safety context, and alignment with current standards of care.
Last Reviewed: April 8, 2026
Short Disclaimer
This information is for educational purposes only and is not medical advice. It does not replace an evaluation by a qualified healthcare professional. For personalized guidance, please schedule a consultation.