Mesenchymal Stem Cell Therapy: A Patient's Guide
You may be reading this because something still isn’t right. Your knee still stiffens after a short walk. Your shoulder lets you sleep only in one position. Your hip feels older than the rest of you. Or perhaps the problem isn’t one joint at all, but a pattern of inflammation, fatigue, immune dysregulation, or slow recovery that never fully resolves.
Many patients reach this point after doing what they were told to do. They’ve tried medications, physical therapy, injections, rest, supplements, and time. Some found partial relief. Few feel fully restored. That’s where mesenchymal stem cell therapy enters the conversation, not as science fiction, and not as a miracle claim, but as a more advanced way of thinking about repair.
A New Era in Cellular Healing and Regeneration
Modern medicine is excellent at crisis care. It can stabilize, replace, suppress, and remove. What it often struggles to do is help injured or inflamed tissue recover in a more coordinated way. A painful joint may get a steroid shot. An autoimmune flare may be quieted with stronger medication. A tendon injury may be monitored until surgery becomes the next step.
Those approaches have value. They just don’t always address the biological environment that allowed degeneration, inflammation, or poor healing to persist.
Mesenchymal stem cell therapy takes a different approach. Instead of focusing only on symptom control, it aims to influence the tissue environment itself. These cells communicate with damaged or inflamed areas, help calm disruptive immune activity, and support the body’s own repair processes. In plain language, they don’t just cover the alarm. They help the body respond more intelligently to the reason the alarm keeps sounding.
That distinction matters to patients who want more than temporary relief. It matters to active adults who want to move with confidence again. It matters to people who aren’t merely trying to avoid decline, but want to preserve function, resilience, and long-term vitality.
A useful way to think about this is to compare conventional symptom care to mopping a wet floor while the pipe above continues to leak. Regenerative medicine asks a better question. Where is the leak, and how do we create the conditions for repair?
At a clinical level, this field has moved well beyond theory. If you want a broader overview of the regenerative philosophy behind these therapies, this guide to cell regeneration therapy offers helpful context.
Mesenchymal stem cell therapy is best understood as a way to improve biological signaling, not as a shortcut around careful diagnosis.
That’s why thoughtful care starts with precision. The right cells, the right patient, the right delivery method, and the right supportive environment all matter. When those pieces align, regenerative medicine becomes less about hype and more about strategy.
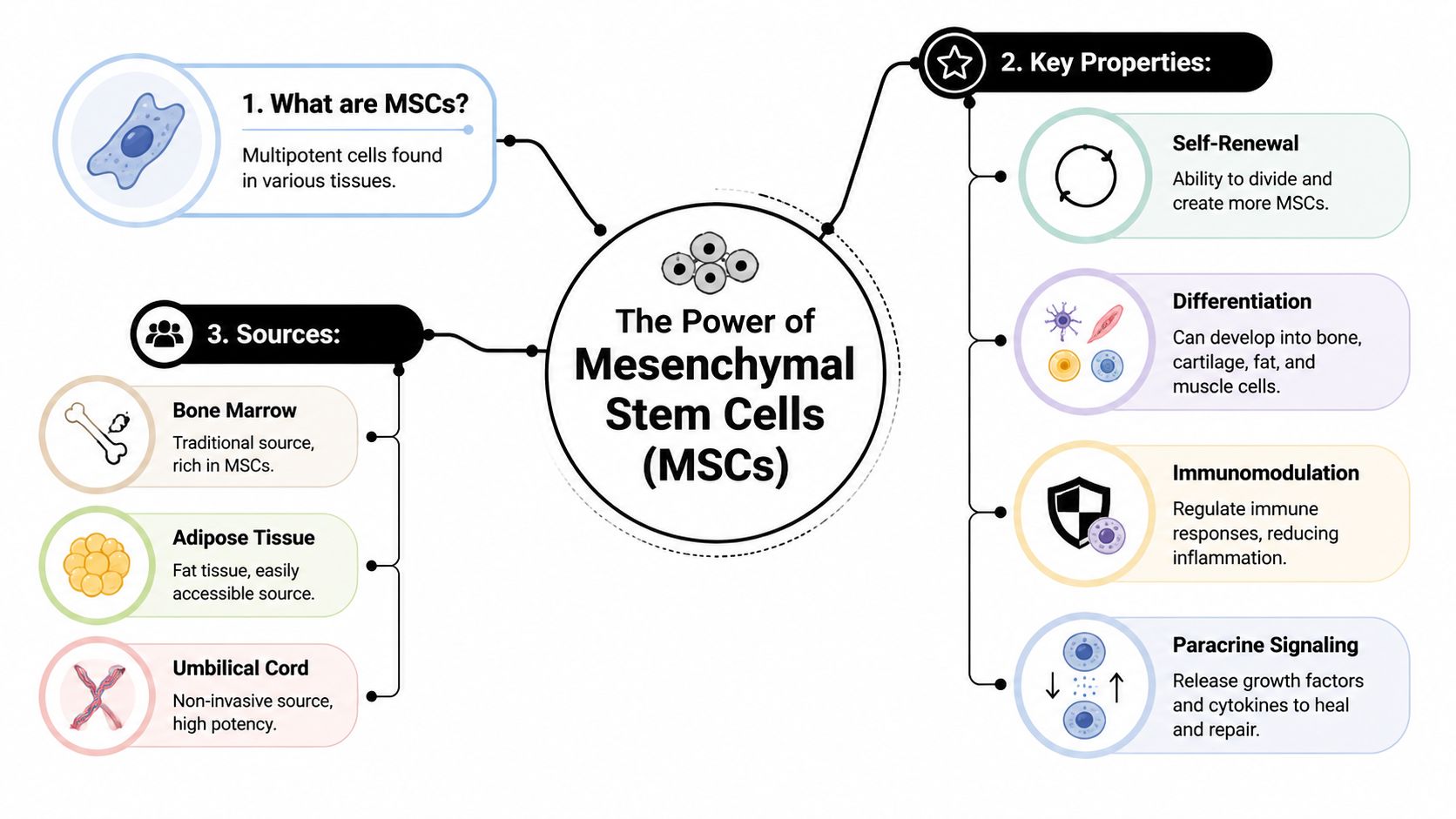
Understanding Mesenchymal Stem Cells and Their Power
The phrase “stem cell” can be confusing because it gets used as if all stem cells are the same. They aren’t. Mesenchymal stem cells, often called MSCs, are a specific category of regenerative cells studied extensively for their ability to support healing, regulate inflammation, and influence tissue repair.
They are often described as the body’s repair coordinators. That description is more accurate than the common image of stem cells turning directly into brand-new body parts. In practice, MSCs behave more like a highly skilled construction management team than a pile of replacement bricks. They arrive, assess the environment, send instructions, reduce chaos, and help organize repair.
What makes MSCs different
MSCs are considered multipotent, which means they can develop along several tissue-supporting pathways rather than every possible cell type in the body. If you’d like a clear explanation of that distinction, this comparison of multipotent vs pluripotent stem cells is useful.
For patients, the more important point is what MSCs do once they are administered. Their value lies in four broad capabilities:
Immune modulation helps calm excessive inflammatory signaling.
Tissue support encourages local repair processes in joints, tendons, cartilage, and other structures.
Cellular communication allows MSCs to influence neighboring cells through biochemical messages.
Regenerative orchestration helps the body shift from a state of ongoing irritation toward one of more orderly healing.
That’s why MSC therapy appears across so many areas of regenerative medicine, from orthopedic injuries to inflammatory and immune-related conditions.
Why allogeneic cells matter
At our clinic, we only use allogeneic MSCs. That means the cells come from carefully screened donor-derived tissue sources rather than being harvested from the patient at the time of treatment. For many patients, that is an important advantage. The goal is to work with potent, clinically prepared cells rather than relying on tissue that may already reflect age, inflammation, or chronic illness.
Our biotechnology lab produces five different types of stem cells, including placental, Wharton’s jelly, adipose, endometrial, and dental pulp sources. Each source has distinct biological characteristics, and physicians select from these options based on the treatment goal, tissue target, and overall clinical plan.
Practical rule: In regenerative medicine, cell quality matters as much as cell quantity.
That quality standard isn’t a vague promise. Clinical-grade MSCs are characterized using International Society for Cell & Gene Therapy criteria, including ≥95% expression of markers such as CD105, CD73, and CD90, with confirmation that the cells can differentiate into bone, cartilage, and fat lineages according to this ISCT-based overview.
What patients should look for in any MSC program
When patients compare clinics, they often focus on the label “stem cells.” That isn’t enough. Better questions include these:
| What to ask | Why it matters |
|---|---|
| Are the cells allogeneic or patient-derived | This affects consistency, potency, and treatment design |
| How are the cells characterized | Proper marker testing helps confirm identity and purity |
| Where are the cells processed | Lab controls influence safety and reliability |
| How is the treatment matched to my condition | Knee arthritis is not the same as autoimmune inflammation |
A strong MSC program is built on sourcing, characterization, clinical judgment, and precise delivery. Without those elements, the term “stem cell therapy” is too broad to be meaningful.
How Mesenchymal Stem Cell Therapy Works in Your Body
Most patients assume stem cells work by settling into damaged tissue and becoming replacement cells. That sounds intuitive, but it’s not the main story. The dominant mechanism in mesenchymal stem cell therapy is paracrine signaling.
That means the cells act through the biological messages they release. Think of them as a mobile pharmacy. They don’t need to stay forever to create an effect. They deliver a concentrated set of instructions that can reduce inflammatory noise and encourage more constructive healing behavior in the tissue around them.
The signaling effect that changes the environment
MSCs release growth factors, cytokines, and extracellular vesicles that influence local cells and immune activity. This signaling can help calm inflammation, support blood vessel formation, reduce cellular stress, and stimulate the body’s own repair responses.
That’s one reason the treatment can feel conceptually different from standard injections. The aim isn’t only to numb pain. The aim is to shift the tissue environment.
A patient with osteoarthritis often finds this confusing. If the cells don’t stay long-term, how can benefits last? Because the therapeutic effect comes from the signals they release. According to multiple human clinical research studies, injected MSCs are often cleared within 48 hours, yet their signaling has been associated with 40 to 60% reductions in pain scores at 12 months in osteoarthritis patients.
Why inflammation control is central
Many chronic conditions share one underlying pattern. The tissue becomes trapped in a loop of irritation, poor recovery, and dysfunctional signaling. In a joint, that may look like swelling, stiffness, cartilage stress, and pain with movement. In systemic inflammatory conditions, it may look more diffuse, such as fatigue, flares, or prolonged immune imbalance.
MSCs help by changing that biochemical conversation.
They calm excessive immune activity rather than blocking all immune function indiscriminately.
They promote repair signals that encourage healthier local behavior from surrounding cells.
They support a transition from a destructive inflammatory state to a more regulated one.
This short video gives a useful visual introduction to that process.
Why delivery method changes the result
How MSCs are given affects how they behave. An IV infusion may be appropriate when physicians want broad systemic immune signaling. A local injection may be preferable when the goal is to target a joint, tendon, or spine-related structure more directly.
Many online explanations become too simplistic. “Stem cells” are not one treatment. They are part of a clinical strategy. A more detailed overview of how stem cell therapy works can help if you want to understand delivery routes and treatment logic in more detail.
The most important therapeutic event may not be the cell becoming tissue. It may be the cell teaching the tissue how to behave differently.
That idea changes how patients interpret results. Improvement may come not from replacement, but from restoration of better signaling, less inflammation, and stronger local repair dynamics.
Clinical Conditions Addressed with MSC Therapy
Patients rarely come in asking for “paracrine signaling.” They come in because they want to walk without pain, train again, sleep through the night, think more clearly, or stop living around flares. The clinical use of mesenchymal stem cell therapy becomes easier to understand when viewed through those goals.
Across regenerative applications, MSC therapies have shown an average success rate of 78%, with about 80% positive outcomes in orthopedic and joint repair applications, according to this summary of clinical results. Those figures don’t guarantee an outcome for any one individual, but they do show why interest has grown among patients dealing with chronic pain and degenerative conditions.
Orthopedic and sports-related conditions
Patients often first encounter MSC therapy when faced with situations like these: a knee with osteoarthritis becomes stiff after sitting, painful on stairs, and swollen after activity. A rotator cuff tear never feels fully stable. Tendonitis improves, then returns with the same movement pattern.
In these situations, physicians use MSCs to help calm the inflammatory environment and support tissue recovery. Common orthopedic targets include:
Knee, hip, and shoulder arthritis where inflammation and structural wear affect movement quality
Meniscus, ACL, and rotator cuff injuries when patients want a regenerative approach within a broader recovery plan
Tendonitis and plantar fasciitis when chronic irritation prevents full healing
Spine-related pain patterns where local inflammation and degeneration contribute to persistent symptoms
For the athlete, the goal may be return to activity. For the active adult, the goal may be a normal walk, a golf swing, or playing with grandchildren without paying for it the next day.
Autoimmune and inflammatory diseases
A second major group includes patients whose primary problem is immune dysregulation rather than a single joint. Rheumatoid arthritis, lupus, multiple sclerosis, psoriasis, Hashimoto’s thyroiditis, Crohn’s disease, and ulcerative colitis all involve a body that is reacting in a way that harms normal tissue.
MSCs are being studied in these settings because of their immunomodulatory effects. That word sounds technical, but the concept is simple. The treatment is designed to help quiet an immune response that has become misdirected or excessive.
For many patients, the question is not “Can this cure my condition?” A better question is “Can this help create a calmer biological environment?” That’s often the more realistic and clinically useful frame. Patients exploring these categories may also benefit from this overview of stem cell therapy for autoimmune and inflammatory diseases.
Some patients seek regenerative care because tissue is damaged. Others seek it because the immune system keeps creating the damage.
Chronic systemic conditions
Then there is the large group of patients who don’t fit neatly into one box. They may describe long COVID, chronic fatigue, fibromyalgia, persistent inflammation, Lyme-related symptoms, post-viral syndromes, asthma, COPD, or immune dysfunction. They often say the same thing in different words: “I don’t bounce back the way I used to.”
These cases require careful evaluation because symptoms can overlap while biology differs. MSC therapy may be considered as part of a broader attempt to reduce inflammatory burden, support repair signaling, and improve resilience. For these patients, treatment planning usually matters more than labels.
Neurodegenerative and neurologic concerns
Patients also ask about Parkinson’s disease, Alzheimer’s disease, and peripheral neuropathy. In these conversations, the role of MSCs is generally explored in relation to inflammation, immune signaling, and supportive regenerative pathways rather than simplistic promises of replacement.
That distinction protects patients from misunderstanding. In advanced conditions, thoughtful physicians focus on goals such as function, support, symptom burden, and quality of life.
Your Personalized Regenerative Protocol
A stem cell procedure by itself is only one part of regenerative care. In real clinical practice, outcomes often depend on the biological setting into which those cells are introduced. If the body is inflamed, poorly oxygenated, sleep-deprived, under-recovered, or metabolically stressed, even excellent cells may be working uphill.
That’s why a modern treatment plan often includes more than one modality. The field itself reflects that momentum. The MSC therapy market was valued at USD 80.9 million in 2024 and is projected to reach USD 296.6 million by 2030 with a 23.5% CAGR from 2025 to 2030, while over 1,200 clinical trials worldwide were investigating MSCs by 2025, according to this mesenchymal stem cell therapy market report. Growth alone doesn’t prove quality, but it does show that regenerative medicine is moving into a more established clinical phase. You can view Longevity Medical Institute's pricing to compare.
Why combination planning matters
A personalized regenerative protocol may combine MSC therapy with supportive tools that enhance signaling, tissue readiness, or recovery capacity.
Some combinations make intuitive sense:
Exosomes may be used to extend or complement cellular communication.
Peptide therapy can provide targeted biological signaling depending on the patient’s goals.
Hyperbaric oxygen therapy may support tissue oxygenation and recovery conditions.
NK cell therapy may be considered in programs focused on immune surveillance and broader longevity strategy.
Each tool does something different. MSCs help coordinate. Exosomes help carry messages. Hyperbaric therapy helps create a more favorable tissue environment. Peptides can reinforce specific repair or recovery pathways.
How diagnostics shape the protocol
Personalization transitions from marketing language to reality. Physicians may look at imaging, functional concerns, inflammatory patterns, recovery history, and laboratory data before deciding whether a patient needs local injection, systemic support, adjunctive therapies, or all of the above.
A clinic such as Longevity Medical Institute integrates physician-led regenerative programs with an in-house clinical lab, full-body MRI capabilities, ultrasound-guided procedures, and complementary therapies such as exosomes, peptides, NK cell protocols, and hyperbaric oxygen therapy. In practical terms, that model allows treatment planning to be based on findings rather than guesswork.
A better way to think about protocol design
A personalized protocol is less like ordering one product and more like conducting an orchestra. One instrument alone may sound good. Coordinated well, the full system produces a far better result.
The question isn’t only “Which cells will you use?” It’s also “What environment are you creating for those cells to do their job?”
That’s the standard patients should expect from serious regenerative care.
The Patient Journey From Consultation to Recovery
For many patients, the science is no longer the biggest mystery. The practical experience is. They want to know what happens, what the treatment day feels like, and how recovery is managed.
The first step is a consultation built around the condition, not just the procedure. A physician reviews symptoms, prior imaging, treatment history, current medications, activity goals, and the larger health picture. In some patients, the problem is straightforward. In others, the visible complaint, such as knee pain, sits on top of broader issues like chronic inflammation, biomechanical overload, or poor recovery capacity.
Step one through step three
The journey usually unfolds in a sequence.
Clinical evaluation
This includes determining whether the patient is an appropriate candidate for mesenchymal stem cell therapy and whether the target is local, systemic, or both.Diagnostics and treatment mapping
Advanced imaging, lab work, ultrasound assessment, or broader wellness diagnostics may be used to clarify where inflammation or degeneration is occurring.Procedure planning
Physicians decide on route of administration, cell type, adjunctive therapies, and recovery instructions based on the diagnosis and patient goals.
Patients often feel calmer once they understand that regenerative medicine should be mapped, not improvised.
What procedure day usually feels like
If treatment includes an IV infusion, patients generally spend the session in a monitored clinical setting. If treatment includes a local injection into a knee, shoulder, hip, or similar structure, imaging guidance matters. This is not just about comfort. It’s about placement.
A key challenge in MSC therapy is getting the cells to the tissue that needs them. According to this review of MSC homing and delivery, systemic IV infusions rely on natural homing signals, while ultrasound-guided local injections can dramatically improve delivery efficiency for orthopedic treatment. That matters because accurate placement can directly influence whether a treatment reaches the intended tissue environment.
Precise delivery is part of the therapy, not a minor technical detail.
For many patients, the local injection itself feels similar to other image-guided procedures they may have had before, though the treatment plan and follow-up are more involved.
The recovery phase patients should expect
Recovery doesn’t always mean immediate pain relief. Some patients notice change early. Others improve gradually as inflammation settles and function returns over time. Activity modification, sleep quality, hydration, nutrition, and rehabilitation all influence how well the biology can respond.
A useful reference for planning is this guide on recovery time after stem cell injection, which helps patients understand why healing timelines are rarely identical.
Common elements of the aftercare phase include:
Follow-up guidance to monitor response and adjust activity
Rehabilitation support when movement quality or strength needs to be rebuilt
Adjunctive therapies if the original plan included hyperbaric sessions, peptides, or exosomes
Expectation management so patients understand that regenerative care supports healing, but doesn’t erase biomechanics, overuse, or neglect
The best patient experience is one where nothing feels hidden. You know why you were selected, why a specific route was chosen, and what your job is after the procedure.
Frequently Asked Questions About MSC Therapy
How do I know if I’m a good candidate
Good candidates usually have a clearly defined condition, realistic goals, and a medical history that supports regenerative treatment planning. The right candidate isn’t determined by enthusiasm alone. It requires diagnosis, review of imaging or labs when appropriate, and a physician’s judgment about whether mesenchymal stem cell therapy fits the problem.
What makes one stem cell clinic better than another
Look for four things. Cell source, lab quality, physician oversight, and delivery precision. Patients should ask whether the clinic uses allogeneic cells, how the cells are characterized, whether processing follows recognized quality standards, and whether procedures such as joint injections are image-guided rather than estimated.
What is the regulatory environment in Mexico
Patients should ask directly about clinical licensing, laboratory standards, and physician supervision. A credible clinic should be able to explain its regulatory framework, quality controls, and how its cells are processed and handled. Clear answers are a good sign. Vague language is not.
How soon can I expect results
Some patients notice changes early, especially in inflammation or comfort. Others improve more gradually as tissue signaling, function, and rehabilitation progress. The timeline depends on the condition being treated, the delivery method, the patient’s baseline biology, and whether other supportive therapies are part of the protocol.
Are exosomes or other adjuncts always necessary
Not always. They may be useful when physicians want to reinforce signaling, improve the tissue environment, or build a broader regenerative program. The need depends on the patient, not a preset package.
If you’re considering Longevity Medical Institute, the next step is a physician-led consultation focused on your diagnosis, goals, and whether mesenchymal stem cell therapy fits your case. The institute publishes educational resources under Treatments & Resources for patients exploring regenerative care in a more informed way.
Author
Dr. Kirk Sanford, DC, Founder & CEO, Longevity Medical Institute. Dr. Sanford focuses on patient education in regenerative and longevity medicine, translating complex therapies into clear, practical guidance for patients.
Medical Review
Dr. Félix Porras, MD, Medical Director, Longevity Medical Institute. Dr. Porras provides clinical oversight and medical review to help ensure accuracy, safety context, and alignment with current standards of care.
Last Reviewed: April 28, 2026
Short Disclaimer
This information is for educational purposes only and is not medical advice. It does not replace an evaluation by a qualified healthcare professional. For personalized guidance, please schedule a consultation.