Are Stem Cells Safe? What Our Published Research Concluded

Stem cell therapy has attracted tremendous attention over the last several years, and for good reason. Across a wide range of diseases and clinical conditions, published research has shown encouraging results, with many studies demonstrating the potential of stem cells. This growing body of research has created real hope for patients seeking options beyond the conventional sick-care model and surgery.
At the same time, hope must always be matched with responsibility.
When patients hear about the potential benefits of stem cells, one of the most important questions they should ask is:
Are Stem Cells Safe?
It is a fair question, and it deserves more than a brief or simplified answer. It deserves a careful review of the published research.
At Longevity Medical Institute, our approach has always been grounded in physician oversight, clinical judgment, careful patient evaluation, and ongoing review of the scientific literature. Several months ago, we decided to take that a step further by conducting a broader and more structured review of the published safety data so we could better understand the landscape and share that perspective more clearly with our patients.
What follows is not promotional language or vague reassurance. It is a discussion grounded in more than 15 years of published clinical data across multiple disease categories, as well as the conclusions drawn from our own formal review of that literature. I believe this is exactly the level of transparency and diligence patients deserve when they place their trust in us.
Are Stem Cells Safe?
Yes. To answer this question, we held ourselves to the same international standards used in published medical research. Five of our LMI physicians, along with our biomedical engineer and clinical chemist, all with experience in clinical research, conducted a formal systematic review of the medical literature to better understand what the evidence actually shows about stem cell safety.
A systematic review is considered one of the highest scientific standards for evaluating a medical topic because it examines the full body of relevant published evidence rather than relying on a single study or opinion.
What Did Our Published Research Show?
The most important finding was clear: the published research did not reveal evidence of serious treatment-related stem cell complications.
Across more than 60 stem cell clinical studies over 15 years, 4000 patients, and multiple disease categories, the evidence did not show increased risk of serious complications such as cancer, infection, thromboembolic events, organ issues, or death attributable to the types of stem cells we administer.
Statistically, that matters. When no serious treatment-related events are observed in a population of approximately 4,000 patients, the estimated true risk is below 0.075% percent.
For our patients, this should be highly reassuring, suggesting that serious treatment-related complications should not be a significant concern based on the published human clinical literature.
Did Anyone Independently Review Our Research?
Yes. Our work was peer-reviewed and published in the Journal of Stem Cell Research International, an independent scientific research journal. As part of that process, an external research team evaluated the methodology, analysis, and conclusions of our work before publication.
In our study, we focused on donor-derived mesenchymal stem cells, often called MSCs. These are the same type of stem cells we administer in clinical practice and produce in our in-house biotechnology lab.
You can read our fully published research here:
"Safety of Allogeneic Mesenchymal Stem Cell Therapy Across Human Clinical Indications: A Systematic Review of Landmark Clinical Trials and Published Safety Evidence"
Do Our Types of Stem Cells Cause Cancer?
No. One of the most common concerns people have about stem cells is whether they could increase the risk of cancer.
Our research specifically examined this question, including the potential for malignant transformation. To date, the available human clinical literature has not shown evidence that the stem cell types we use are associated with an increased risk of cancer. No confirmed cases of treatment-related malignancy from these administered stem cell types have been reported in human clinical trials, and pooled safety analyses have not identified a higher cancer risk among treated patients.
This distinction is important because not all stem cells are the same. Some stem cell types, particularly pluripotent stem cells such as embryonic stem cells, have the ability to develop into almost any tissue in the body. Because of that unrestricted developmental potential, they carry a known risk of forming tumors. We don’t use pluripotent stem cells, so don’t worry!
Our stem cells are fundamentally different. They are multipotent mesenchymal stem cells (MSCs) whose primary role is to regulate inflammation and support tissue repair. In clinical use, MSCs have not demonstrated a reproducible association with cancer formation.
Taken together, the current body of human evidence supports allogeneic MSC therapy as a well-tolerated and scientifically promising regenerative medicine platform with a favorable safety profile. Allogeneic simply means the cells come from carefully screened healthy donors.
In our biotech lab, our stem cells are derived from placental, umbilical cord, adipose, endometrial, and dental pulp tissues, rather than being taken from the patient’s own body (aka autologous stem cells).



Did Our Research Include Conditions Those Treated at LMI?
Yes. The studies we reviewed covered a wide range of medical conditions, many of which overlap with the types of conditions treated at Longevity Medical Institute. These include heart disease, autoimmune disorders, bowel diseases, lung injury, orthopedic conditions such as osteoarthritis, kidney disease, and inflammatory disorders.
We were particularly interested in donor-derived stem cells because that’s what we make in our biotech lab and administer to our patients.
How Did Your Research Evaluate Safety?
In the studies we analyzed, we carefully looked for serious complications that could be linked to stem cell therapy. These included cancer development, organ damage, severe immune reactions, blood clots, infections, or death.
Researchers also tracked more common but less severe reactions that may occur shortly after treatment, allowing them to distinguish between mild expected responses and true safety concerns.
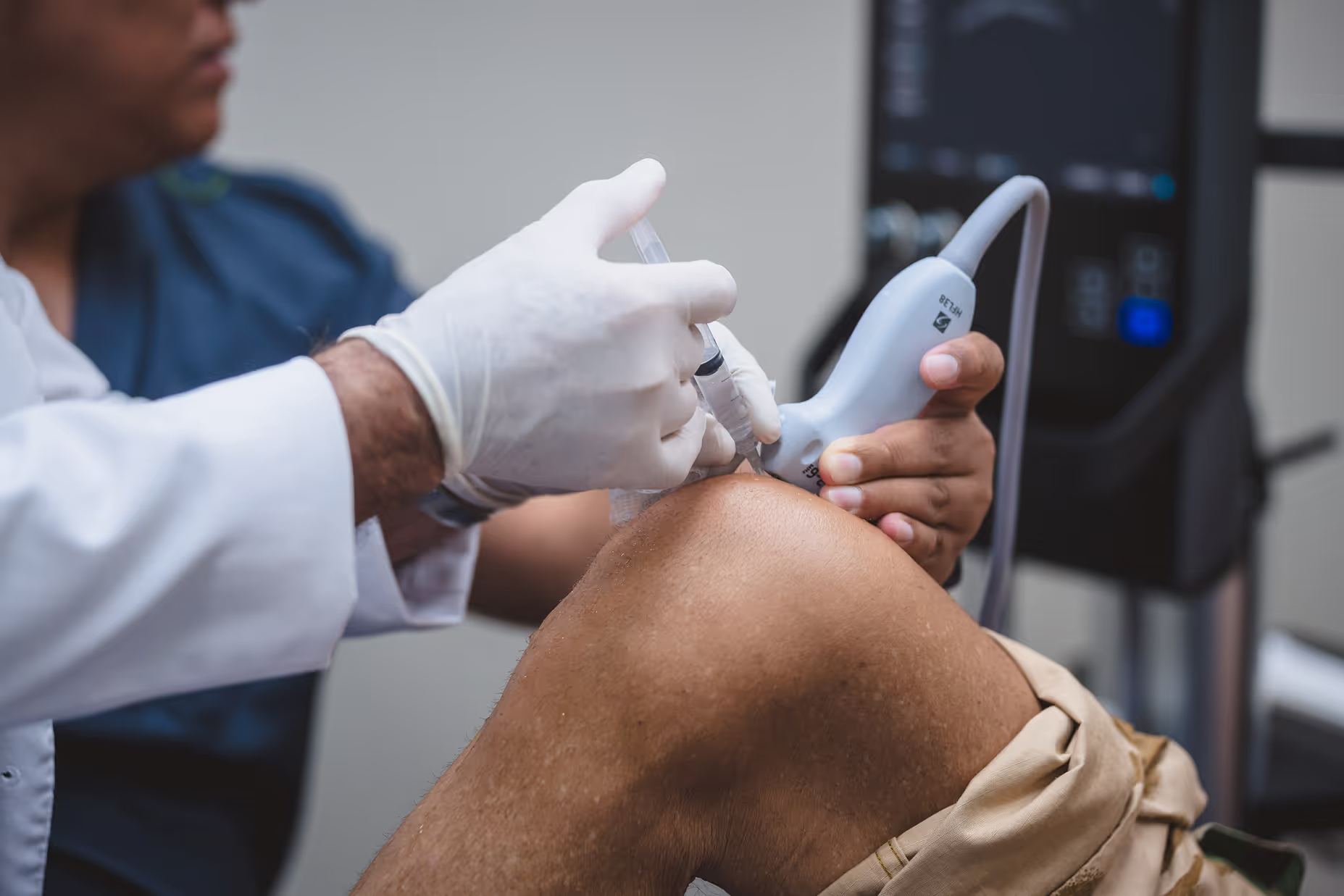
Were Both IV Stem Cells and Injection Therapies Studied?
Yes. Stem cells have been studied using multiple delivery methods. Clinical trials have evaluated intravenous infusions, direct joint injections, targeted injections into damaged tissues, and specialized delivery techniques used in cardiac treatments. Despite these different administration methods, the safety profile remained consistent across studies.
Were There Any Reported Reactions to Stem Cell Administration?
Yes. Reported reactions to stem cell administration were most commonly mild and transient. A small percentage of patients, roughly 5 percent, experienced temporary fever, fatigue, or chills shortly after treatment. When present, these reactions were generally short-lived and resolved without significant intervention.
Why does this happen?
Mesenchymal stem cells are biologically active and begin interacting with the immune system soon after administration. Rather than remaining inactive, they communicate with immune cells, inflammatory mediators, and repair pathways throughout the body.
As a result, some patients may experience a brief immune-related response, such as low-grade fever, fatigue, or chills. In most cases, this is not considered harmful and reflects the body’s normal recognition of and response to the therapy.
This distinction is important. A temporary, self-limited reaction is not the same as a serious treatment-related complication.
Overall, the literature suggests that in 1 out of every 20 patients, the most commonly reported reactions to mesenchymal stem cell administration are mild, manageable, and short-lived.

Want to Know More About How We Are Leading Stem Cell Research?
Mexico has one of the largest concentrations of stem cell clinics in the world, yet relatively little clinical research is published from within the country. That gap matters.
We are committed to advancing the field through meaningful scientific contributions, including publication in internationally recognized research journals.

What This Means for You as a Patient
I often say that trust begins with safety and transparency. In previous newsletters, I have shared what sets us apart, including our distinction as the only federally COFEPRIS-licensed stem cell clinic and biotechnology lab in Los Cabos. I have also discussed the rigorous laboratory processes behind our therapies, including flow cytometry analysis used to characterize the cells produced in our biotechnology lab. If you would like to learn more about that process, you can revisit our March newsletter titled 250 Billion Cells Every Day.
Over the past several years, research programs around the world have continued to explore how stem cells may help support recovery and tissue repair in a wide range of conditions.
Current human research, including over 1,500 clinical studies, suggests that the most promising applications for stem cell therapy include musculoskeletal conditions, inflammatory and autoimmune disorders, metabolic diseases, liver disease, frailty and age-related decline, neurodegenerative disorders, sexual wellness conditions, and respiratory diseases.
While these discoveries are advancing the field of regenerative medicine, they also reinforce an important point: as the science grows, so does the responsibility to deliver these therapies safely and under proper medical oversight.
We have now answered another critically important question about the safety of stem cells, one that every patient should care about.
As with any medical therapy, it is important that treatment be performed by qualified physicians who follow established clinical protocols and safety standards.
At Longevity Medical Institute, our commitment is to combine rigorous science, careful clinical protocols, and transparency so that patients can make informed decisions about their health.
Our stem cell therapies are designed to support the body’s natural repair mechanisms and may be used as part of a comprehensive treatment approach for a wide range of conditions, including:
• Age-related concerns
• Musculoskeletal injuries and degenerative conditions
• Inflammatory and autoimmune disorders
• Metabolic conditions
• Neurodegenerative disorders
• Organ-related illnesses
• Respiratory conditions
• Sexual wellness and hormonal concerns
Want to learn more about the conditions we treat? Click Here
Missed a previous newsletter? You can view it here.
Only Two Weeks Left to Take Advantage of Our Stem Cell Special!
To celebrate the opening of our new Stem Cell Biotechnology Laboratory, we are offering a limited-time promotion for March only.
Purchase a two stem cell treatment package and receive one additional stem cell treatment at no cost.
Price: $3,500 (plus IVA), that's a savings of $1,750! Guided ultrasound injection is an additional cost of $450 per region.
This allows you to complete a three-treatment regenerative series while only paying for two!
All packages must be purchased during the promotional period in March. Once purchased, treatments must be used within twelve months.
To reserve your stem cell package or schedule your consultation on a protocol, please contact Ivanna or Ariana, and they will be happy to assist you.
Your Feedback Makes a Difference
We're proud to share that we've already received multiple five-star reviews — thank you to everyone who's taken the time to support us!
As a new clinic, your positive feedback not only means the world to us, but it also helps boost our visibility online so more people can discover us.
If you’ve had a positive experience at Longevity Medical Institute, we’d be truly grateful for your review. Select the button below to Leave a Google Review
See more of our articles
Be up to date and check our newest educational materials.

Sleep Medicine and In-Residence Medical Services Now Available!
From the beginning, my goal for Longevity Medical Institute® has been to build a truly integrative and comprehensive model of longevity care. As we continue to evolve that model, there are times when one strategic addition can strengthen multiple areas at once.

Inside Mexico’s Premier Longevity Clinic: The Future of Healthcare at Longevity Medical Institute®
Tendencia Magazine sat down with Dr. Kirk Sanford, Founder and CEO of Longevity Medical Institute®, one of Mexico’s leading longevity and stem cell clinics, for a conversation on the future of regenerative medicine, the evolving meaning of longevity, and the new standard of health optimization.

I'm Not Sure I Want to Know
Over the past few months, since launching our MRI program in December, I’ve had numerous conversations with both patients and prospective patients.
Bridging the expertise of traditional medicine with the innovations of tomorrow.
With decades of experience spanning tens of thousands of patient cases, our doctors provide an unparalleled depth of care.