Is Stem Cell Therapy in Mexico Safe? A Clinical Guide
You’re probably reading this after seeing two very different stories.
One says Mexico is a smart place to access advanced regenerative medicine that isn’t easily available in the United States or Canada. The other warns that “stem cell tourism” can be risky, poorly regulated, and full of inflated promises. Both stories contain part of the truth.
The key question isn't whether stem cell therapy in Mexico is safe. The better question is whether the specific clinic, laboratory, physician team, and treatment process you’re considering are safe.
That distinction matters. A medically supervised, licensed, physician-led program operating within Mexico’s regulatory framework is very different from a sales-driven clinic using vague language, unclear sourcing, and no meaningful follow-up. Patients often lump both into the same category. They shouldn’t.
The Critical Question Behind Your Search for Healing
Most patients who ask, “Is Stem Cell Therapy in Mexico Safe?”, are not casually browsing. They’re dealing with something real.
It may be knee pain that keeps returning despite injections and physical therapy. It may be a shoulder injury that limits training and sleep. It may be chronic inflammation, fatigue, or an autoimmune condition that hasn’t responded the way they hoped. Many are also frustrated by how narrow their options feel at home.
What I see most often is a patient caught between hope and caution. They want access to advanced care. They also don’t want to make an expensive or unsafe mistake.
That’s a healthy instinct.
A careful patient usually starts by reading broad overviews such as this page on stem cell therapy in Mexico, then realizes the search gets murky fast. One clinic emphasizes luxury. Another emphasizes lab science. Another makes sweeping promises that sound too good to be true. Safety becomes hard to judge because the marketing language often looks polished across the board.
Practical rule: Safety isn’t determined by the country name in the headline. It’s determined by licensing, laboratory standards, physician oversight, informed consent, and aftercare.
That’s why a simple yes or no answer doesn’t help enough.
Stem cell therapy in Mexico can be safe. It can also be poorly handled when patients choose clinics that aren’t operating to a serious medical standard. The difference comes down to what can be verified, not what can be advertised.
If you approach this decision the way you’d approach any high-level medical procedure, with documentation, questions, and a low tolerance for vague answers, you put yourself in a much stronger position.
Understanding Mexico's Regulatory Framework for Stem Cells
A patient can land in Mexico, walk into two clinics on the same day, and see polished websites, white coats, and confident promises at both. One may be operating with documented clinical controls and proper laboratory procedures. The other may be relying on marketing language that sounds medical but does not hold up under scrutiny.
That distinction matters more than the country itself.
Mexico regulates cell-based medicine through COFEPRIS, the Federal Commission for Protection against Sanitary Risks. For a patient, that means there is a legal and administrative structure for authorizing health facilities, overseeing sanitary compliance, and setting expectations for how human tissues and cells are handled. The question is narrower and more practical. Is the specific clinic you are considering operating within that structure, and can it prove it?
What COFEPRIS oversight actually means
Patients often hear COFEPRIS described as Mexico’s counterpart to the FDA. The comparison is useful at a high level, but it does not answer the question a careful patient should ask. What documents, standards, and operating procedures sit behind the treatment being offered?
A legitimate clinic should be able to explain its approvals clearly and match them to its actual workflow. That includes how cells are sourced, processed, stored, transported, and administered. It also includes who is responsible at each step, from laboratory personnel to the treating physician.
Mexico also uses official standards, known as NOMs, to govern health and manufacturing practices. In stem cell care, patients will often hear references to standards related to good manufacturing practices and the handling of human cells and tissues. The exact code matters less to a patient than the clinic’s ability to show how those standards are applied in daily operations, not just mentioned in a sales call.
In practical terms, a credible program should be ready to answer four direct questions:
Where are the cells processed, and under what quality controls?
How is sterility protected from collection through administration?
Which physician is responsible for the treatment plan and informed consent?
What records can the clinic provide to verify compliance, licensure, and lab procedures?
If those answers are vague, delayed, or framed as proprietary, caution is warranted.
Regulation only protects patients when the clinic follows it
This is the trade-off I want patients to understand. A country can have a real regulatory structure, and individual clinics can still fall short of serious medical practice. I have reviewed cases where patients were shown general certificates, photos of lab equipment, or broad claims about regenerative medicine, yet received little clarity on chain of custody, release testing, physician oversight, or follow-up responsibility.
That is why due diligence has to move past slogans.
Start with the existence of regulation. Then verify clinic-level compliance inside that system. A useful next step is to review what a biotechnology stem cell lab in Mexico should show you about processing standards, quality control, and clinical integration.
The reassuring point is straightforward. Mexico has a formal framework for stem cell medicine. The safety difference comes from whether a clinic can document that it meets that framework consistently, case by case.
The Superior Safety Profile of Allogeneic Stem Cell Therapy
Not all stem cell approaches carry the same clinical profile. That’s especially important if you’re comparing modern, physician-led programs with older models or heavily restricted approaches.
At serious regenerative centers, the discussion should focus on cell source, processing quality, sterility, viability, and delivery protocol. Those factors shape safety far more than broad marketing labels.
Why allogeneic therapy matters
For this discussion, one point should be clear. We’re talking about allogeneic therapy, not patient-harvested cells.
Allogeneic mesenchymal stem cells are sourced from donated tissue and prepared under controlled laboratory conditions. In premium clinical settings, that approach removes the need to subject the patient to a harvest procedure before treatment. From a patient experience and procedural safety standpoint, that’s a meaningful advantage.
COFEPRIS-licensed clinics should be able to provide documentation of their license that clearly displays the clinic’s name. These licenses are issued to a specific clinic and cannot be sublicensed or sponsored by another facility.
For patients, the practical takeaway is straightforward. A well-prepared allogeneic product allows treatment to begin with a standardized cell source instead of adding another medical procedure to obtain cells from the patient first.
What makes an allogeneic program safer
When an allogeneic protocol is done well, its safety profile depends on discipline.
That means:
Ethical sourcing: The clinic should be able to explain where the cells come from and how consent and donor screening are handled.
Laboratory controls: Processing has to occur in a setting built around sterility, traceability, and quality standards.
Physician-directed selection: The doctor should decide whether the patient is an appropriate candidate, not a sales coordinator.
Clear route of administration: Joint injection, IV therapy, or combined protocols should be chosen for a clinical reason, not because they sound more advanced.
This is also where patients should be wary of broad claims about “the most powerful cells” or “universal cure” language. The better clinics usually speak more carefully. They talk about suitability, expected response, procedural planning, and monitoring.
Quality of the cells is part of safety
Patients often focus only on whether a procedure will hurt or whether a country is safe to travel to. Those are reasonable concerns, but they miss the core issue.
The biologic product itself is part of the safety equation.
If cell handling is inconsistent, if viability is poorly documented, if storage practices are sloppy, or if the source is hard to verify, the risk profile changes immediately. That’s true even if the procedure room looks impressive.
This is why any serious evaluation of Is Stem Cell Therapy in Mexico Safe has to include the laboratory side of the conversation. A good starting point is understanding the standards discussed in is stem cell therapy safe, especially as they apply to cell sourcing, handling, and physician oversight.
The safest regenerative procedure starts long before the injection. It starts with the quality of the cells and the controls surrounding them.
That’s the distinction informed patients should care about most.
Navigating Potential Risks and Unregulated Tourism
A balanced answer has to acknowledge risk directly.
Stem cell therapy is not automatically safe because a clinic has a polished website, a luxury waiting area, or a location in a popular medical tourism corridor. The highest-risk scenarios usually come from clinics that cut corners, blur regulatory boundaries, or sell treatments for nearly everything without a meaningful diagnostic process.
Where problems usually begin
The major hazards tend to follow a familiar pattern.
A clinic may be unclear about licensing. It may offer treatment after only a brief phone call. It may avoid detailed discussion of laboratory standards. It may use emotionally charged language such as “miracle,” “guaranteed,” or “works for everyone.”
Those are not minor marketing issues. They’re signs that the clinic may be operating more like a retail funnel than a medical practice.
One of the biggest problems for patients is the lack of independent, third-party safety data. Regulated clinics often emphasize certifications, but there is still minimal aggregated outcome data or complication data from neutral sources. That gap makes patient diligence even more important.
Risks patients should understand
A thoughtful clinic should discuss the possibility of known procedural and biologic risks rather than pretending they don’t exist.
These include:
Infection risk: If sterility standards are weak, the procedure becomes less safe immediately.
Immune reactions: Poor processing or poor product quality can increase the chance of unwanted responses.
Ineffective treatment: Not every patient responds, and not every condition is an appropriate target.
Financial and emotional harm: A poorly selected treatment can cost time, money, and momentum in your broader care plan.
If a clinic talks only about upside and never discusses limitations, you’re not hearing a medical conversation. You’re hearing a sales conversation.
The phrase patients should be careful with
“Stem cell tourism” gets used broadly, and sometimes unfairly. Traveling for medical care isn’t the problem by itself. Many patients travel for excellent orthopedic, dental, surgical, and regenerative care.
The problem is unregulated tourism, where the travel experience is polished but the clinical foundation is weak.
That’s why discerning patients should ask for evidence of standards instead of reassurance by branding. If the answers stay vague, move on.
The safest patients are rarely the ones who feel least worried at the beginning. They’re the ones who turn concern into a disciplined screening process.
Your Vetting Framework Green Flags vs Red Flags
The fastest way to lower your risk is to stop asking, “Do I like this clinic?” and start asking, “Can this clinic document the standard of care it claims to provide?”
That shift changes everything.
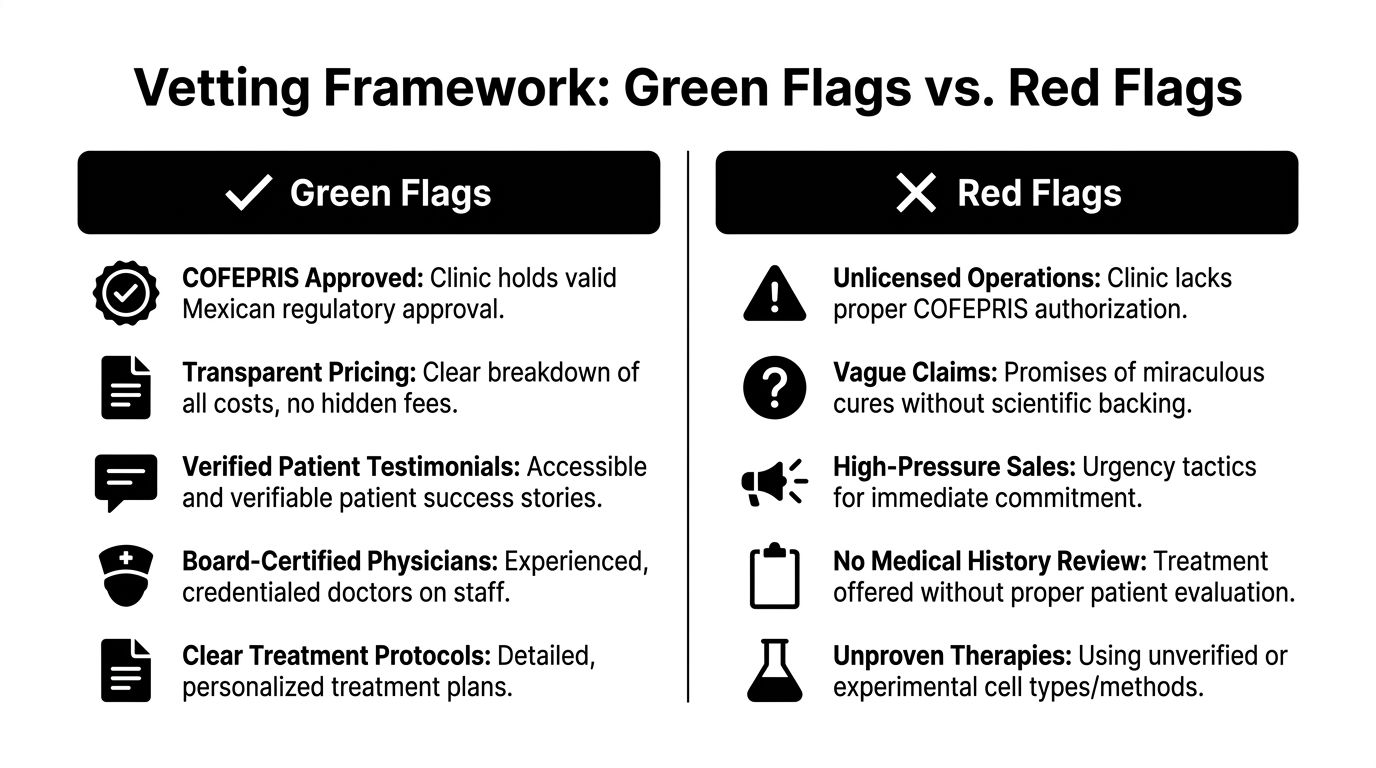
Green flags that deserve your attention
Stem cell therapy in Mexico achieves strong safety profiles in COFEPRIS-certified clinics when proper licensing and GMP standards are followed. Facilities holding key licenses for cell application, storage, and manufacturing, while adhering to standards such as NOM-059-SSA1-2015, can mirror U.S. safety levels.
Our peer-reviewed research, Safety of Allogeneic Mesenchymal Stromal Cell Therapy Across Human Clinical Indications, further supports this. Across multiple clinical areas and major trials, allogeneic MSC therapy demonstrates a strong safety profile, with no consistent evidence of increased risk for serious adverse events, infection, malignancy, or treatment-related mortality. The most commonly observed effect has been mild, transient fever.
Together, this reinforces a simple point: safety in stem cell therapy is driven by proper regulation, clinical standards, and correct application.
That gives patients a useful benchmark. You’re looking for a clinic that behaves like a medical institution, not a wellness storefront.
Look for these signs:
Verifiable regulatory approvals: The clinic should be willing to show its COFEPRIS-related documentation and explain what those approvals cover.
Laboratory transparency: Ask where cells are processed, what standards govern the lab, and how sterility and handling are controlled.
Physician-led evaluation: Your records, diagnosis, imaging, medications, and goals should be reviewed before anyone recommends treatment.
Condition-specific discussion: The doctor should talk about your knee, shoulder, spine, autoimmune issue, or recovery goals specifically, not generically.
Realistic language: Good clinics discuss candidacy, possible benefit, limitations, and follow-up. They don’t promise cures.
Documented treatment plan: You should receive a clear explanation of what’s being administered, how, and why.
One example of the kind of resource patients often use during this stage is a guide to choosing a licensed stem cell clinic in Mexico, which can help translate regulatory language into practical screening criteria.
Red flags that should stop the conversation
Some warning signs are immediate disqualifiers.
Treatment offered before chart review: If nobody has asked for records or imaging, they aren’t practicing personalized medicine.
Vague sourcing: If the clinic can’t clearly explain what cells are being used and how they’re handled, that’s a problem.
One protocol for everyone: A serious clinic won’t present the same package to an athlete with a rotator cuff injury and a patient with inflammatory symptoms.
Pressure to pay quickly: Urgency is useful in sales. It’s dangerous in medicine.
Broad cure claims: The more conditions a clinic claims to cure with certainty, the more skeptical you should become.
No aftercare structure: If the clinic’s involvement ends when you leave the building, the model is incomplete.
What to ask: “Who determines candidacy, where are the cells processed, what documentation can you provide, and what follow-up do you require after treatment?”
Clinic Vetting Checklist
| Vetting Criteria | Green Flag (High Standard of Care) | Red Flag (Potential for Risk) |
|---|---|---|
| Regulatory status | Clinic can explain and verify licensing and compliance | Licensing language is vague or evasive |
| Medical evaluation | Physician reviews records and determines candidacy | Sales team recommends treatment immediately |
| Cell source | Source is explained clearly and ethically | Source is unclear, inconsistent, or avoided |
| Lab quality | Sterility, handling, and quality controls are discussed plainly | Lab details are hidden behind marketing terms |
| Treatment planning | Protocol is personalized to diagnosis and goals | Same package is pushed for many conditions |
| Claims | Outcomes are discussed conservatively | “Cure” and “guarantee” language is used |
| Follow-up | Aftercare and monitoring are built into care | No structured follow-up is offered |
| Financial communication | Pricing is clear and documented | Deposits are pushed before medical review |
A strong clinic doesn’t just sound credible. It holds up under detailed questions.
The Non-Negotiable Role of Physician Consultation and Aftercare
The consultation is not a formality. It’s one of the strongest safety filters in the entire process.
A patient considering stem cell therapy should expect a real medical review. That means history, diagnosis, past treatments, imaging when relevant, medication review, goals, limitations, and a candid discussion about whether the treatment is appropriate at all.
Why broad claims fall apart in real medicine
One of the major unanswered issues for patients is the long-term efficacy and safety for their specific condition. Clinics often lack comparative data for different cell types in orthopedic or autoimmune applications, which is exactly why a deep physician consultation matters.
That point deserves emphasis.
A patient with knee arthritis, a patient with lupus, and a patient recovering from an ACL injury should not receive the same conversation. Their risk profile, goals, and expected response are different. So is the standard for what counts as success.
What a meaningful consultation should include
A serious consultation usually covers several areas:
Diagnostic clarity: The physician should define what problem is being treated.
Candidacy: Not everyone is a suitable candidate. The doctor should say so when appropriate.
Expected outcomes: You need realistic goals, not inflated hope.
Procedure planning: Route, setting, and support therapies should have a clinical rationale.
Recovery and monitoring: Follow-up should be discussed before treatment, not after.
One resource patients can review when comparing care models is how physician-led stem cell therapy in Mexico differs from transactional clinic models.
Personalized medicine is safer medicine. The more your treatment plan reflects your records, your condition, and your goals, the less likely you are to be treated like a commodity.
Aftercare is part of safety
Aftercare is often overlooked because patients focus on the procedure day. But good aftercare protects the patient in several ways.
It creates a channel for reporting unexpected symptoms. It gives the clinical team a way to assess early response. It also helps set reasonable expectations about when improvement may or may not appear.
A clinic that treats the procedure as the entire product is thinking too narrowly. Safety includes what happens before treatment, during treatment, and after you return home.
A Guide for US and Canadian Medical Travelers
For patients traveling from the United States or Canada, safety also depends on planning the trip well. Medical travel feels much easier when the logistics are clear before you leave home.
What to organize before you travel
Start with paperwork and communication.
Bring your recent medical records, imaging reports if your condition is orthopedic or neurologic, medication list, and a written summary of prior treatments. If a clinic asks for these in advance, that’s usually a good sign. It means the physician intends to review your case properly.
You should also know, in writing:
What procedure is planned
How long you’re expected to stay
What support is available after treatment
Who to contact if questions come up once you’re back home
Travel itself should feel coordinated, not improvised. Patients coming to San José del Cabo often appreciate having a single point of contact who can help with scheduling, arrival planning, and post-visit communication.
What to expect financially and legally
Patients should assume they’re paying privately unless told otherwise. Cross-border regenerative medicine typically requires direct payment arrangements rather than standard insurance processing.
You should also expect informed consent documents that explain risks, limitations, and the experimental or emerging nature of certain applications where relevant. If consent is rushed, minimized, or handled casually, pause.
For patients who want a visual overview of what the travel and treatment experience can look like, this short video is a helpful reference point.
Travel should support recovery, not complicate it
The most practical plans are the safest ones.
Don’t schedule treatment in the middle of a packed vacation itinerary. Give yourself room for evaluation, procedure day, and a calm recovery window. If you’re receiving treatment for pain, inflammation, mobility limits, or fatigue, build your travel schedule around your body, not around sightseeing.
Our clinic model is designed to provide a structured, high-level patient experience, integrating physician oversight with on-site diagnostics and laboratory support. Whether you choose our facility or another, the principle remains the same. Medical travel should be organized, clinically grounded, and thoroughly documented from the initial consultation through follow-up care.
Making Your Decision with Confidence and Clarity
So, is stem cell therapy in Mexico safe?
It can be. In the right setting, with the right oversight, it can be a responsible and well-structured medical choice. In the wrong setting, it can become an expensive and avoidable risk.
The difference isn’t marketing quality, destination appeal, or how confident a coordinator sounds on the phone. The difference is whether the clinic can document high standards in regulation, laboratory practices, physician leadership, informed consent, and follow-up care.
That’s the framework that matters.
If you remember only a few points, remember these. Verify the clinic’s compliance. Ask direct questions about cell source and lab handling. Expect a true physician consultation. Be wary of cure claims. And don’t confuse a luxury experience with a medical standard of care.
Patients usually feel more confident once they stop trying to answer the question in broad terms. “Is Mexico safe?” is too vague to guide a medical decision. “Is this clinic transparent, licensed, physician-led, and prepared to care for me properly?” is a much better question.
That is how careful patients make good decisions.
Author
Dr. Kirk Sanford, DC, Founder & CEO, Longevity Medical Institute. Dr. Sanford focuses on patient education in regenerative and longevity medicine, translating complex therapies into clear, practical guidance for patients.
Medical Review
Dr. Félix Porras, MD, Medical Director, Longevity Medical Institute. Dr. Porras provides clinical oversight and medical review to help ensure accuracy, safety context, and alignment with current standards of care.
Last Reviewed: April 13, 2026
Short Disclaimer
This information is for educational purposes only and is not medical advice. It does not replace an evaluation by a qualified healthcare professional. For personalized guidance, please schedule a consultation.
If you’d like help evaluating whether a stem cell program in Mexico meets the standards outlined above, Longevity Medical Institute offers physician-led consultations focused on candidacy, safety, and treatment planning for patients seeking regenerative care in Los Cabos.