Unlocking Hope: Stem Cells in Multiple Sclerosis Treatment
Living with multiple sclerosis (MS) means navigating a condition where your own immune system mistakenly attacks the central nervous system. The experience is deeply personal and often unpredictable. While traditional treatments have made huge strides in managing symptoms, many people are looking for ways to do more than just manage—they want to fundamentally shift the course of their health.
This is where regenerative medicine opens up a new conversation. Instead of only treating the symptoms of MS, we are exploring how allogeneic stem cells in multiple sclerosis can work on a deeper level to rebalance the body’s own systems and support its incredible capacity for repair.
A New Horizon for Multiple Sclerosis
The journey with MS is unique to every single person, marked by an individual set of symptoms and an unpredictable path. Conventional treatments are vital for slowing the disease and managing relapses, but they often leave people searching for more proactive ways to restore function and reclaim their health.
This is where the principles of regenerative medicine introduce a compelling new direction. Instead of only addressing the downstream effects of MS, advanced therapies work to influence the underlying biological processes driving the disease.
Shifting the Focus from Management to Repair
The core issue in MS is an autoimmune response that damages myelin, the protective sheath around your nerve fibers. This damage disrupts the communication lines between the brain and the body, leading to a wide range of neurological symptoms. The goal of using stem cells for MS is to intervene directly in this destructive cycle.
At Longevity Medical Institute, our focus is on using allogeneic mesenchymal stem cells (MSCs) sourced from ethically screened, donated umbilical cord tissue. These cells are remarkable for their ability to:
Modulate the immune system: They help calm the overactive immune response that causes the damage in the first place.
Promote a healing environment: They release powerful signaling molecules that encourage the body's own repair mechanisms to get to work.
Protect neural tissue: They provide vital support to existing nerve cells, helping to shield them from further injury.
This approach represents a shift from simply managing a chronic condition to actively supporting the body’s potential for restoration and resilience. By introducing potent, healthy signaling cells, the aim is to create an internal environment that is less inflammatory and more conducive to healing.
Our purpose is to translate this sophisticated science into safe, personalized treatments that are clear and accessible. As you explore this guide, you will gain a deeper understanding of how this therapy works. To learn more about the foundational concepts, you can check out our guide on how regenerative medicine works. We are committed to empowering you with the knowledge to make confident decisions about your health, helping you pursue a greater quality of life and reclaim a sense of vitality.
To understand how stem cell therapy works for Multiple Sclerosis, it's helpful to stop thinking of the cells as simple replacements for damaged tissue. Instead, picture them as a highly intelligent biological repair crew.
At Longevity Medical Institute, we work exclusively with allogeneic mesenchymal stem cells (MSCs), which are ethically sourced from donated and meticulously screened umbilical cord tissue. These are not just any cells; they are young, vibrant, and possess a powerful ability to communicate with and direct your body’s own systems.
Think of these MSCs as a combination of diplomatic peacekeepers and master project managers. Their primary job isn’t just to rebuild what’s broken, but to fundamentally change the environment, calming the autoimmune conflict so the body's own healing processes can finally get to work.
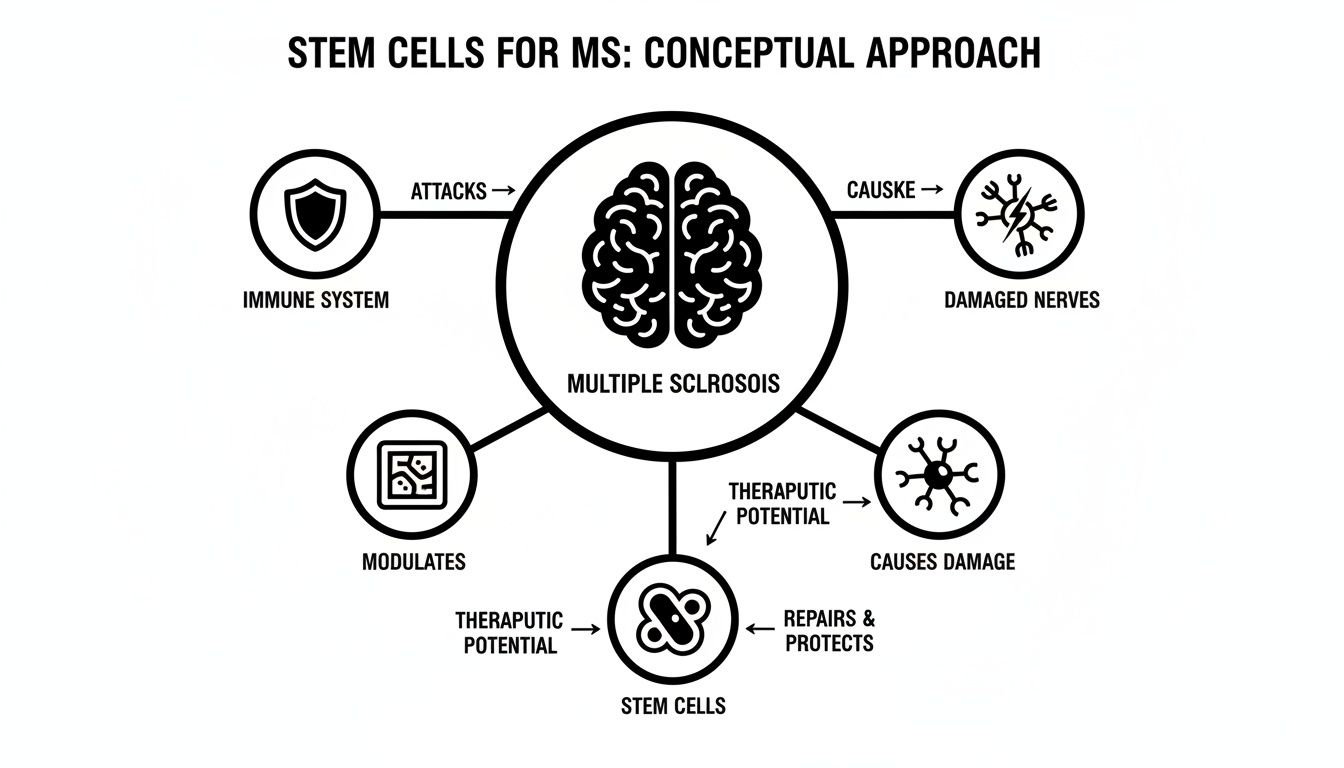
The real insight here is that these cells orchestrate a coordinated response. They work to simultaneously address both the misguided immune attack and the neurological damage it leaves behind—a crucial strategy for managing a complex disease like MS.
While our clinical focus is on allogeneic MSCs, it's helpful to know about the different types of stem cells being explored in MS research. Each has a unique role and therapeutic target.
Types of Stem Cells Used in Multiple Sclerosis Research
| Cell Type | Source | Primary Mechanism | Therapeutic Goal in MS |
|---|---|---|---|
| Mesenchymal Stem Cells (MSCs) | Umbilical Cord, Bone Marrow, Adipose Tissue | Immunomodulation & Trophic Support: Calm immune attacks and release protective growth factors. | Reduce inflammation, protect nerve cells, and encourage the body's own repair processes. |
| Hematopoietic Stem Cells (HSCs) | Bone Marrow, Peripheral Blood | Immune System "Reset": Eradicate the existing dysfunctional immune system and rebuild it with healthy cells. | Halt inflammatory activity in highly active, relapsing forms of MS. |
This comparison highlights why allogeneic MSCs are so promising for a broad range of MS cases. Their primary goal is not to replace the immune system, but to intelligently guide it back toward a state of balance.
The Three Pillars of Action
The therapeutic power of allogeneic MSCs in MS is driven by three core functions that work together to disrupt the disease's progression. Understanding these actions makes it clear why this is such a promising frontier in medicine.
Immunomodulation: Calming the Immune System
In MS, the body's immune system gets its signals crossed and begins attacking the protective myelin sheath around your nerves. Allogeneic MSCs are exceptional immunomodulators. They release powerful signals that "re-educate" these overactive immune cells, encouraging them to stand down and dialing back the inflammatory assault. It’s like a skilled negotiator stepping in to de-escalate a conflict, restoring peace where there was chaos.Trophic Support: Protecting Nerve Cells
Beyond simply calming the immune attack, MSCs actively protect nerve cells through what’s known as trophic support. They secrete a rich cocktail of growth factors and neuroprotective molecules that act like a biological shield, defending vulnerable neurons from further damage. This creates a supportive microenvironment where nerve cells can not only survive but function better.Promoting Regeneration: Signaling for Repair
MSCs also act as powerful directors, signaling the body’s own resident repair systems to wake up and get to work. They don't necessarily become new nerve cells themselves. Instead, they encourage the natural repair processes that have been overwhelmed by the chronic inflammation of MS. This helps create an environment where remyelination—the rebuilding of that damaged myelin sheath—can actually happen. You can learn more about the source of these potent cells in our guide to umbilical cord stem cell therapy.
This approach of using young, allogeneic MSCs provides a consistent, potent, and high-quality therapeutic tool designed to interrupt the disease process without the limitations of using a patient’s own cells, which are older and may carry the same autoimmune dysfunction.
Global research continues to reinforce these mechanisms. Mesenchymal stem cell therapies are gaining significant traction, particularly for progressive forms of MS. One notable trial in Italy, for instance, showed profound neuroprotective effects, with higher doses being linked to greater preservation of brain tissue over time. Researchers found the cells helped reduce inflammation and even reprogrammed key immune cells from a damaging state to a protective one. You can read more about these groundbreaking MS stem cell research findings to see the scientific momentum for yourself.
Taking a Clear-Eyed Look at the Evidence for MS Stem Cell Therapy
When you're exploring any advanced therapy for multiple sclerosis, cutting through the hype to see the actual scientific evidence is non-negotiable. For patients and their families looking into the potential of stem cells for MS, understanding the real state of clinical research provides both a sense of hope and a much-needed dose of perspective.
Our goal here isn’t to sell you on a miracle. It’s to give you a realistic picture of where the science stands today, translating the findings from key clinical trials into insights that matter for you. We focus specifically on the data surrounding the allogeneic mesenchymal stem cells (MSCs) we use, because our entire approach is built on a foundation of transparency and trust.
Key Findings from Clinical Research
Across the board, the growing body of research shows that allogeneic MSCs have a very strong safety and tolerability profile. In one trial after another, patients generally experience few serious side effects, making it a solid platform for continued study. But the more compelling story is in the signals of efficacy—the measurable, real-world improvements suggesting the therapy is having a positive impact.
These are the kinds of signals we’re seeing in the research:
Improved Mobility: Some studies report patients making tangible gains in their walking speed and overall endurance.
Enhanced Bladder Function: A notoriously challenging MS symptom, bladder control has shown marked improvement in some trial participants.
Reduced Brain Atrophy: Using advanced MRI analysis, some researchers have observed a slowing of brain volume loss, which is a key marker of disease progression.
This evidence points toward a therapy that could do more than just manage symptoms; it appears to have the potential to fundamentally change the course of the disease. The research focus is shifting from simply slowing MS down to actively intervening in its biology. This is a game-changer, especially for progressive forms of MS where options have always been so limited.
Our approach to treating autoimmune conditions with stem cells is detailed further in our guide to stem cell therapy for autoimmune and inflammatory diseases.
A Global Effort with Exciting Data
The sheer scale of the research happening right now is a powerful indicator of the field’s momentum. An incredible number of clinical trials are underway worldwide, reflecting a shared, global commitment to unlocking the benefits of stem cell therapy for MS.
As of early 2024, there are over 900 clinical trials investigating therapies for MS across the globe, with hundreds actively recruiting or in progress. One particularly bright spot comes from a Phase II study involving intrathecal injections of MSCs. It showed significant improvements in walking ability and, remarkably, a 69% improvement in bladder function compared to placebo.
These exciting results are now paving the way for larger, later-stage trials. To see the data for yourself, you can learn more about these encouraging MS stem cell trial results.
While the journey from a clinical trial to a standard, everyday medical practice is a long one, these outcomes provide tangible hope. They demonstrate that stem cell therapy holds real promise for slowing disease progression and, most importantly, improving the daily functions that truly define a person's quality of life.
At Longevity Medical Institute, we are guided by this evolving science. Our commitment is to provide access to therapies that are not only on the leading edge but are also supported by a growing foundation of solid clinical evidence. This allows us to build personalized treatment plans that are both forward-thinking and firmly grounded in safety and realistic expectations.
Determining if You Are a Candidate for Therapy
Deciding if allogeneic stem cell therapy is the right path for your multiple sclerosis journey is a significant and deeply personal step. It’s a decision that can only be made through a careful, collaborative evaluation between you and our clinical team.
This therapy is not a universal solution; it is a specialized medical intervention for individuals who meet specific criteria. This ensures we can maximize both safety and the potential for a positive outcome.
At Longevity Medical Institute, our entire philosophy is built on this principle of precise, personalized medicine. The ideal candidate for our stem cells in multiple sclerosis protocol is often someone living with either a relapsing-remitting or progressive form of MS.
Typically, these are individuals who have already tried conventional treatments but haven’t achieved the level of disease control or quality of life they are searching for.
The Comprehensive Evaluation Process
To determine if this therapy is right for you, we conduct a comprehensive evaluation designed to build a complete picture of your health. This meticulous process is the cornerstone of responsible and effective care.
Our assessment includes:
A thorough review of your medical history: We take a deep dive into your MS diagnosis, the course of your disease, past treatments, and your overall health journey.
Analysis of recent MRI scans: Your latest imaging is crucial. It allows us to assess current disease activity, lesion load, and any changes in brain volume.
Evaluation of your current functional status: We look closely at your mobility, cognitive function, and how MS currently impacts your daily life, often using standardized measures like the Expanded Disability Status Scale (EDSS).
This in-depth analysis gives our medical team the data needed to confirm that regenerative therapy is a medically sound option for your specific situation.
Understanding Who Is Not a Candidate
Just as important as identifying ideal candidates is being completely transparent about who is not. These are specific conditions, or contraindications, where stem cell therapy may not be safe or appropriate.
Patient safety is our highest priority. We believe the most successful outcomes are built on a foundation of trust, transparency, and an unwavering commitment to "do no harm." This is why our screening process is so rigorous.
Contraindications for allogeneic stem cell therapy may include:
Active, uncontrolled infections: Your body must be free of significant infections before undergoing any regenerative therapy.
A history of certain cancers: A detailed review of your oncological history is required to ensure complete safety.
Severe, unstable health conditions: Unmanaged cardiac, pulmonary, or kidney disease can increase procedural risks and must be carefully considered.
Pregnancy or breastfeeding: The effects of this therapy during pregnancy are unknown, so it is strictly contraindicated.
Our commitment to a personalized, data-driven assessment ensures that we only move forward when we have a strong, evidence-informed reason to believe in a positive and safe outcome. For a deeper dive into our safety-first approach, you can explore our detailed overview on whether stem cell therapy is safe for conditions like MS.
Your Regenerative Journey With Us
Choosing to embark on a regenerative treatment plan is a significant decision. We believe the entire experience should feel as restorative as the therapy itself. Your journey with us is a seamless, physician-led process designed around your unique biology and clinical needs, all while prioritizing your personal comfort. It is a partnership built on trust, transparency, and an unwavering commitment to excellence.
From your initial consultation, our focus is squarely on you. We move beyond one-size-fits-all protocols to create a therapeutic strategy that is meticulously aligned with your specific health goals. This is the foundation of everything we do.
Phase 1: Personalized Preparation
Your path to regeneration begins with a detailed consultation with one of our specialist physicians. This is a comprehensive discovery process where we take the time to listen to your MS history, understand your objectives, and begin to map out a potential course of action.
This initial conversation is followed by advanced diagnostics to create an incredibly detailed picture of your current health. This data-rich approach allows our medical team to design a stem cell protocol that is not just personalized, but precise.
Comprehensive Lab Work: We analyze key biomarkers to assess everything from systemic inflammation to immune function, giving us a clear picture of your overall health.
Advanced Imaging: AI-enhanced MRI scans allow us to evaluate disease activity with exceptional detail, providing a crucial baseline to measure progress against.
Based on this wealth of data, our medical team collaborates to create your bespoke treatment plan, ensuring your therapy is both safe and optimized for your individual biology.
Phase 2: The Treatment Procedure
The administration of stem cells in multiple sclerosis at our institute is a calm and expertly managed medical procedure, performed in a state-of-the-art, sterile clinical environment designed for your comfort and safety.
The procedure itself is a minimally invasive intravenous (IV) infusion. Here’s a simple breakdown of what you can expect on the day:
Preparation: You’ll be settled into one of our private treatment suites. Our medical staff will then prepare the IV line, which is a quick and simple process.
Infusion: The allogeneic mesenchymal stem cells are slowly introduced into your bloodstream through the IV drip. The entire process is painless and generally takes one to two hours.
Monitoring: Throughout the infusion, our dedicated medical team will be right there with you, monitoring you closely to ensure your comfort and well-being.
Recovery: After the infusion is done, there is a short observation period before you’re cleared to leave the clinic and relax for the rest of the day.
We use only the highest-quality cells from our fully licensed and certified biotechnology lab, ensuring every dose meets stringent standards for viability and purity. You can learn more about our advanced biotechnology stem cell lab in Mexico and our commitment to quality.
Our focus on using allogeneic, umbilical cord-derived stem cells means you receive a potent, vibrant, and consistent therapeutic product, administered with the highest degree of clinical precision and care.
Phase 3: Post-Treatment Care And Progress Monitoring
Your journey with us extends far beyond the day of your procedure. We believe that robust follow-up care is fundamental to achieving the best possible outcomes, which is why our team designs a detailed schedule to monitor your progress and provide continuous support.
This phase often includes supportive therapies designed to enhance the effects of the stem cells and promote overall wellness. Depending on your individual needs, this might involve hyperbaric oxygen therapy to boost cellular energy or personalized physical medicine to improve functional mobility.
For our many international patients, our seamless concierge services manage all travel and accommodation logistics. Our team ensures your entire experience in beautiful San José del Cabo is premium, stress-free, and focused solely on your health and recovery. From the moment you arrive until you depart, we are here to support you.
Setting Realistic Goals and Measuring Success
When you decide to pursue stem cell therapy for multiple sclerosis, one of the most important first steps is to have a frank conversation about what success really looks like. It is critical to understand that this is a disease-modifying therapy, not an instant cure. The goal is to partner with your body’s own systems to create meaningful, lasting improvements.
We focus on ambitious but achievable outcomes: slowing down the progression of MS, calming the systemic inflammation that drives the disease, improving neurological function, and ultimately, giving you a better quality of life. This is a progressive therapy, and the timeline for results is as unique as you are.
Defining Your Personal Success
Success isn’t a single, universal benchmark. It’s a collection of personal victories that signal a real shift in your health. For some, the first signs might be subtle—a bit more energy in the afternoons, sharper thinking, or less of that crushing fatigue. For others, the changes may be more visible functional gains.
Together, we’ll look for a range of potential improvements, including:
Better motor control and coordination.
Gains in mobility, balance, and how fast you can walk.
A reduction in spasticity or muscle stiffness.
Improved bladder or bowel control.
These benefits often appear gradually over several months as the stem cells work to modulate your immune system and create an environment that encourages your body’s own repair mechanisms to get to work.
How We Measure Progress Together
Your personal experience—how you feel day-to-day—is the single most important measure of success. We then combine your feedback with objective data to build a complete and accurate picture of your progress.
This allows us to track every change with precision and refine your long-term wellness plan. Our toolkit includes:
Functional Tests: We use standardized assessments like timed walks (such as the 25-Foot Walk Test) and other neurological exams to get concrete data on your mobility and coordination.
Objective Clinical Data: We keep a close eye on follow-up lab work that tracks inflammatory markers. We also use advanced MRI scans to look for changes in lesion activity or even brain volume over time.
Success is a collaborative process of setting achievable goals and carefully tracking progress. It’s about celebrating every step forward, whether it’s a subtle shift in daily energy or a measurable improvement in physical function.
While our institute focuses exclusively on allogeneic MSCs, the powerful potential of stem cell-based approaches is being demonstrated across the entire field. For example, data from major studies on hematopoietic stem cell transplantation (HSCT) for aggressive MS is incredibly encouraging.
In those trials, two-thirds of patients had no disability worsening four years after treatment. Some patient groups even saw remission rates as high as 70%, underscoring the profound ability of stem cells to halt the neurodegenerative process. You can read more about these impressive HSCT trial outcomes to better understand the scientific momentum in this area.
Your Questions About Stem Cell Therapy for MS, Answered
Making the decision to explore advanced therapies for multiple sclerosis is a significant one, and it’s natural to have important questions. This section is designed to give you clear, straightforward answers to the most common concerns we hear from patients, so you can move forward with confidence and clarity.
We believe an informed patient is an empowered one. Our commitment is to full transparency, helping you understand both the science and the process.
Is Allogeneic Stem Cell Therapy for MS Safe?
Patient safety is the absolute foundation of everything we do. The process begins with the cells themselves. The allogeneic mesenchymal stem cells (MSCs) we use are sourced exclusively from licensed, accredited laboratories that follow strict international guidelines for screening donated umbilical cord tissue. This ensures the cells are rigorously tested for purity, potency, and freedom from any contaminants.
The therapy itself is a minimally invasive intravenous (IV) infusion, performed by our specialist medical team in a controlled, sterile clinical environment. While any medical procedure has potential risks, allogeneic MSCs have consistently shown an excellent safety profile in clinical trials for autoimmune conditions like MS. Serious side effects are exceptionally rare, with most patients only reporting temporary, low-grade fatigue.
How Long Does It Take to See Results?
The timeline for seeing results is unique to every person. The therapy works by progressively calming the immune system and creating a healing environment, so the changes you feel will be gradual, not instantaneous.
Some patients tell us they notice initial shifts in their energy, mental clarity, or spasticity within the first few weeks to months. More significant functional improvements—like better mobility or coordination—often develop more slowly over the following six to twelve months as your body continues to respond. We track this journey with you through consistent follow-up appointments and functional assessments to measure your progress.
The goal is sustainable, long-term improvement. The cells initiate a cascade of healing signals, and the full scope of benefits unfolds over time as your body’s systems respond to this new, healthier environment.
Why Do You Use Allogeneic Donor Cells for MS?
At Longevity Medical Institute, we exclusively use allogeneic (donor) MSCs for treating autoimmune conditions like multiple sclerosis, and the reasoning is both scientific and practical. This approach offers critical advantages.
First, these cells come from the umbilical cords of healthy, stringently screened newborns, meaning they are incredibly young, vibrant, and possess maximum therapeutic power. They haven't been affected by the aging process or a lifetime of environmental exposures.
Second, using a patient’s own cells for an autoimmune disease introduces a theoretical risk of re-introducing immune cells that carry the same dysfunctional programming driving the condition in the first place. Allogeneic MSCs don't have this problem. Instead, their powerful immunomodulatory properties help “re-educate” your own immune system. This gives us a standardized, high-quality, and potentially more effective tool for managing stem cells in multiple sclerosis.
Author
Dr. Kirk Sanford, DC — Founder & CEO, Longevity Medical Institute. Dr. Sanford focuses on patient education in regenerative and longevity medicine, translating complex therapies into clear, practical guidance for patients.
Medical Review
Dr. Félix Porras, MD — Medical Director, Longevity Medical Institute. Dr. Porras provides clinical oversight and medical review to help ensure accuracy, safety context, and alignment with current standards of care.
Last Reviewed: March 29, 2026
Short Disclaimer
This information is for educational purposes only and is not medical advice. It does not replace an evaluation by a qualified healthcare professional. For personalized guidance, please schedule a consultation.