Stem Cell Therapy Mexico vs USA: A 2026 Patient Guide
A lot of patients arrive at the same point before they ever book a consultation. They’ve tried physical therapy, injections, medications, rest, supplements, and time. They’re still dealing with knee pain, shoulder dysfunction, back flare-ups, autoimmune symptoms, or a level of fatigue that keeps narrowing daily life.
Then they start comparing options. In the United States, they often find limited access, narrow treatment pathways, and pricing that puts advanced regenerative care out of reach. In Mexico, they find broader availability, lower pricing, and a flood of conflicting information. That’s where confusion sets in. Some clinics are excellent. Some are not. The question isn’t whether one country is better than the other. The core question is where a patient can access the right therapy, under the right oversight, with the right clinical standards.
The Crossroads of Regenerative Medicine Choosing Your Path
A typical patient considering Stem Cell Therapy Mexico vs USA isn’t looking for a shortcut. They’re looking for a realistic path forward.
It may be the active adult with knee osteoarthritis who doesn’t feel ready for joint replacement. It may be the former athlete with a rotator cuff injury that never fully healed. It may be the professional who has spent years managing inflammation, pain, and loss of function while hearing that regenerative medicine sounds promising, but not for them, not here, not now, or not without a very large out-of-pocket cost.
That moment matters. Patients often assume that looking outside the U.S. means settling for less. In practice, it can mean making a better-informed decision about access, cell quality, and treatment design.
A physician-led clinic in Mexico can operate at a very high clinical standard while offering therapies that are difficult or unavailable to access in the U.S. That distinction matters most for patients seeking allogeneic mesenchymal stem cells, especially when they want a treatment plan built around orthopedic recovery, inflammation modulation, or long-term function rather than a limited trial framework.
Patients who do best in this process usually stop asking, “Which country is cheaper?” and start asking, “Which clinic can show me exactly what it uses, how it’s regulated, and who is responsible for my care?”
The strongest decisions come from clarity, not urgency.
If you’re still building a foundation for what regenerative care involves, it helps to start with a practical overview of how regenerative medicine works. Once you understand the treatment logic, the Mexico versus U.S. question becomes much easier to evaluate.
What patients are really choosing between
This isn’t a simple choice between domestic care and medical travel.
It’s usually a choice between:
- Limited access versus broader access to regenerative protocols
- Higher cost versus greater affordability
- Narrow regulatory pathways versus more flexible clinical application
- Variable clinic quality in both countries, which means the clinic matters more than the passport stamp
That last point is the one patients often miss.
Mexico is not automatically better. The U.S. is not automatically safer. Superior care comes from strong standards, transparent sourcing, physician oversight, and clear follow-up.
Understanding the Regulatory Divide: FDA vs COFEPRIS
A patient with knee degeneration comes to our clinic after being told in the U.S. that the only realistic options are pain management, injections with limited biologic potential, or surgery later. Then the same patient starts reading about cultured allogeneic MSCs in Mexico and asks the right question: why is one option available there and not easily available here?
The answer starts with regulation, but patients need more than a simple “FDA versus COFEPRIS” summary.
In the United States, the FDA generally treats advanced stem cell products as biologic drugs. That classification brings strict requirements for manufacturing, characterization, approval pathways, and clinical use. The goal is caution and standardization. The practical result is narrower access, especially for therapies that involve expanded or cultured allogeneic cells outside formal trials or limited channels.
That system has real strengths. It can reduce variability and push manufacturers toward high documentation standards. It also slows availability for patients who are dealing with pain, tissue loss, inflammatory conditions, or declining function now, not years from now.
Mexico uses a different regulatory model. COFEPRIS oversees regenerative medicine through licensed facilities, laboratory controls, and medical practice rules that can allow broader clinical use of allogeneic mesenchymal stem cells, including cultured cells, when the clinic and lab meet the required standards. For the right patient, that difference is not theoretical. It changes what treatment options are on the table.
As a physician, I tell patients to avoid two bad assumptions. U.S.-based care is not automatically the best fit. Mexico-based care is not automatically lower quality.
The issue is how the clinic operates inside its regulatory environment.
Mexico’s stem cell sector has grown under that structure. Grand View Research’s outlook on the Mexico stem cells market reports USD 160.0 million in revenue in 2023 and projects USD 369.8 million by 2030, with a 12.7% CAGR from 2024 to 2030. Growth alone does not prove quality, but it does suggest expanding infrastructure, more licensed activity, and greater investment in labs, processing standards, and physician-led programs.
That last point matters.
A treatment can be legal, regulated, and clinically appropriate in Mexico without being broadly available in the United States. Patients often mistake “not FDA available” for “unsafe,” and that is too simplistic. The better question is whether the clinic can show you exactly how it sources, tests, stores, and administers the cells, and which physician is accountable for the plan.
Patients who want to examine the lab side before choosing a clinic should review how a biotechnology stem cell lab in Mexico operates.
Here is what I advise patients to verify before they rely on any claim about regulation:
- COFEPRIS licensing status
- Laboratory processing and quality controls
- Cell sourcing, donor screening, and traceability
- Physician oversight from evaluation through follow-up
- Clear documentation of what is being administered
Regulation sets the boundaries. Quality of care depends on what the clinic does within them.
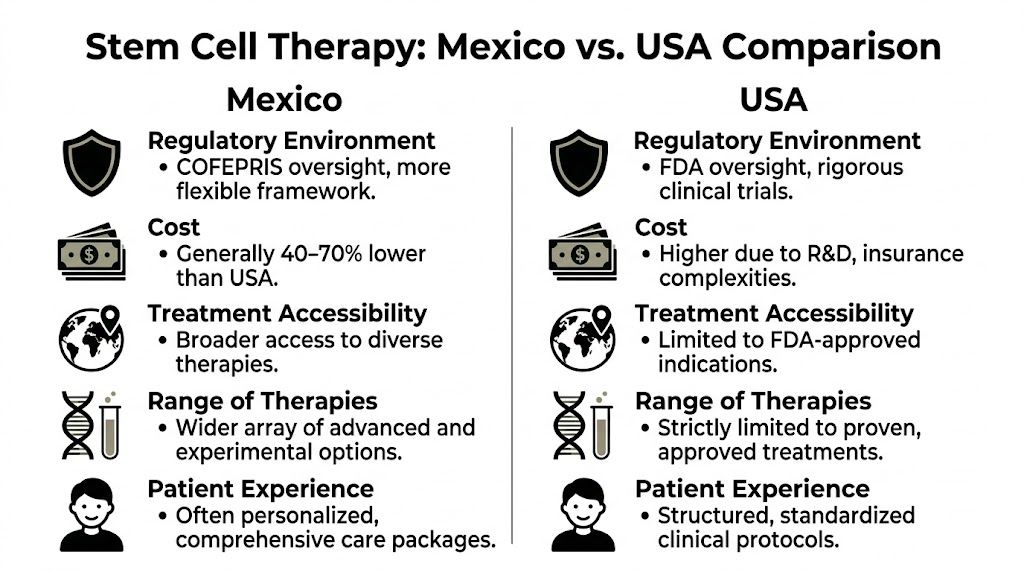
Comparing Stem Cell Therapy in Mexico and the USA
A patient often reaches this section after hearing two very different messages. In the United States, they are told the treatment they want is limited, unavailable, or only offered in narrow settings. In Mexico, they see clinics advertising broader options, lower prices, and faster scheduling. The decision is not whether one country is good and the other is bad. The decision is which setting gives you access to the right cell product, under credible medical oversight, with standards you can verify.
That distinction matters because patients are often comparing different treatment categories, not the same therapy in two locations.
CriterionMexico (at a COFEPRIS-Licensed Clinic)USA (within FDA Guidelines)Regulatory frameworkCOFEPRIS oversight allows broader clinical application of allogeneic MSCs at licensed facilitiesFDA oversight is more restrictive and often limits access outside narrow pathwaysCell approachCultured allogeneic MSCs may be available through licensed regenerative programsAccess is more limited under stricter regulatory constraintsTreatment scopeBroader use for orthopedic, inflammatory, and degenerative presentations in clinical practiceMore restricted availability and narrower useCost profileLower pricing is commonly available in qualified settingsHigher pricing is common due to regulatory and operational burdenClinic variationExcellent clinics exist, but patients must verify licenses, lab standards, and physician oversightStrong controls exist, but access and therapy scope are often narrower
Patients who want a broader clinical overview before comparing clinics can review this guide to stem cell therapy in Mexico.
Cell source and potency
From a physician’s perspective, this is often the biggest practical difference.
In many U.S. settings, access to expanded allogeneic mesenchymal stem cells is limited by regulation. As a result, patients are often offered other biologic options, smaller-scale cell procedures, or no treatment at all unless they enter a formal trial or a very specific pathway. In Mexico, licensed clinics may have access to cultured allogeneic MSCs, including cells derived from umbilical cord tissue, which allows treatment plans built around consistency, dosing, and repeatable manufacturing standards.
That changes the discussion in the exam room.
A cultured allogeneic product is not automatically better because it is offered in Mexico. It is better only when the clinic can document donor screening, expansion methods, release testing, storage conditions, and physician control over how the cells are used. In a serious program, the value is not just access. It is access to a more standardized cellular product than many patients can obtain in the U.S.
Treatment scope
The second difference is clinical range.
In Mexico, licensed regenerative programs may treat orthopedic injuries, degenerative joint disease, some inflammatory presentations, and selected chronic conditions that are harder to access in the United States outside narrow regulatory channels. In the U.S., many physicians work under tighter restrictions, so even experienced clinicians may have fewer tools available for the same patient profile.
Broader access does not mean broader approval for everyone. Good physicians still exclude patients every week. Advanced joint collapse, untreated instability, major mechanical problems, unrealistic expectations, or the wrong diagnosis can make stem cell therapy a poor choice regardless of country.
That is one of the easiest ways to spot a serious clinic. It gives you a treatment recommendation only after deciding whether you are a candidate.
Lab standards shape the result
Patients naturally focus on the injection, infusion, or procedure day. The treatment outcome starts much earlier.
The lab determines whether the product is traceable, clean, and consistent enough to use with confidence. In a high-level Mexico program, I expect clear answers about tissue sourcing, donor qualification, cell expansion, sterility testing, viability checks, identity markers, cryostorage, chain of custody, and transport from lab to bedside. If a clinic cannot explain those steps in plain language, the lower price stops being an advantage.
For patients comparing Mexico and the USA, this is the insider point many miss. The better Mexico clinics do not compete by cutting corners. They compete by pairing broader legal access with physician-led protocols and disciplined cell manufacturing that many U.S. patients cannot reach in routine practice.
Delivery matters as much as the product
Even an excellent MSC preparation can be used poorly.
Orthopedic treatment requires an accurate diagnosis, good imaging, proper target selection, and technical skill during delivery. The same applies to systemic protocols. Patient selection, dosing logic, timing, route of administration, and follow-up planning all affect whether the treatment is sensible. A clinic that talks only about how many cells it offers, without discussing why those cells are being used in that patient and that tissue, is leaving out the medical part.
We tell patients to compare the entire care model, not just the vial.
What patients should take from the comparison
The strongest Mexico option is often not the cheapest clinic or the one with the most aggressive advertising. It is the clinic that combines access to cultured allogeneic MSCs with transparent lab controls, licensed oversight, and a physician who knows when regenerative treatment fits and when it does not.
The strongest U.S. option may offer excellent medical oversight, but patients often face narrower treatment access, fewer cell-based options, and higher costs for care that is not always equivalent in biologic potency or scope.
That is the practical difference patients need to understand. Stem Cell Therapy Mexico vs USA is a comparison of access, cell type, manufacturing discipline, and clinical judgment. Geography matters. Verified quality matters more.
Navigating Safety and Quality in Regenerative Medicine
A patient flies to Mexico after being quoted a fraction of the U.S. price, only to realize on arrival that no physician has reviewed the MRI, no one can explain how the cells were prepared, and the treatment plan looks identical to every other patient’s. That is the risk patients should focus on.
Safety is determined by process, oversight, and clinical judgment. Country alone does not answer that question.
From an insider’s view, the best clinics in Mexico combine broader access to advanced cell products with controls that serious patients should expect from any high-level medical practice. That matters most for cultured allogeneic MSC programs, where product quality, donor screening, storage, release testing, and physician selection all affect whether treatment is appropriate and whether the biologic has a reasonable chance to perform as intended.
Patients who want a practical framework can start with this guide on whether stem cell therapy in Mexico is safe, then verify each clinic’s standards one by one.
What to ask a clinic before you travel
A credible clinic should answer direct questions clearly and in writing.
Ask for:
- Clinic licensing details so you can confirm the facility is operating under the proper legal and medical framework
- Laboratory information including where the cells are processed, stored, and released for clinical use
- Cell sourcing details covering donor eligibility screening, tissue source, and handling procedures
- Quality control records such as sterility testing, viability reporting, identity markers, and lot release standards when available
- Physician involvement including who reviews your case, who performs the procedure, and who is responsible for follow-up care
Vague answers are a warning sign. So is pressure to pay before your medical questions are answered.
What quality looks like in practice
Quality is visible when a clinic can document how it works.
In a strong program, candidacy is determined after chart review, imaging review when relevant, and a discussion of realistic goals. The cells are not described as a miracle product. They are treated as one part of a medical plan. For cultured allogeneic MSC treatment, that plan should include clear sourcing standards, controlled expansion methods, release criteria, and storage protocols. If a clinic cannot explain those steps in plain language, it should not be trusted with advanced biologic care.
This is one practical advantage of a physician-led, U.S.-standard clinic operating in Mexico. Patients can access treatment pathways that are often harder to obtain in the United States, while still expecting documented protocols, traceable product handling, and direct physician accountability.
What to remember: High standards show up in records, case selection, informed consent, and follow-up planning.
Red flags patients should not ignore
Some problems are easy to spot once you know what to check.
- Everyone is accepted without meaningful review of diagnosis, imaging, or medical history
- Sales staff answer medical questions while the treating physician stays out of reach
- No documentation is available for cell source, testing, handling, or release procedures
- The same protocol is sold to every patient regardless of condition, severity, or treatment goals
- Outcome claims sound guaranteed instead of careful and condition-specific
Good regenerative medicine includes restraint. A serious clinic will turn some patients away, recommend a different approach when needed, or explain why the expected benefit is too limited to justify treatment.
This short video gives patients another way to think about clinic standards and decision-making before treatment:
The role of physician-led care
Physician involvement matters because stem cell treatment is not just a product purchase. It is medical care.
Diagnosis comes first. Imaging often matters. Functional assessment, prior treatment history, contraindications, and post-procedure planning matter too. In my view, patients make better decisions by looking past marketing language and asking who is responsible for the case from evaluation through follow-up.
The best clinics in either country are physician-directed. In Mexico, that model can give patients access to higher-potency cultured allogeneic MSC options without giving up the structure, discipline, and accountability they would expect from a serious medical practice.
Analyzing the Cost and Value of Treatment
A patient calls two clinics for the same diagnosis and gets two very different quotes. One sounds surprisingly cheap. The other feels shockingly high. The question is not which number looks better. The question is what that price is buying.
Mexico usually offers lower treatment costs than the United States, but cost alone does not tell you whether a case is being handled well. In practice, the price gap often reflects differences in overhead, staffing structure, facility costs, and the regulatory path used to bring specific cell products into care. From a physician’s standpoint, that matters because two treatments listed under the same label can differ sharply in cell type, cell dose, processing standards, and clinical planning.
That is why I advise patients to compare value first.
Why lower pricing can still reflect high-level care
A lower price in Mexico can coexist with strong medical standards, especially in physician-led clinics built to U.S. expectations. The better centers use that cost advantage to make advanced care more accessible, not to cut corners.
The practical advantage is straightforward. A clinic operating in Mexico may be able to offer cultured allogeneic MSC protocols that are difficult to access in the United States, while still maintaining disciplined evaluation, documented quality controls, and direct physician oversight. For the right patient, that combination can produce a better treatment option at a more reasonable total cost.
Price still needs context.
A low quote may exclude imaging review, procedure planning, physician follow-up, or the specific cell preparation that influenced your decision to seek care. A high quote may reflect real quality or high U.S. overhead costs.
What patients should compare
A useful cost comparison looks beyond the invoice and into the treatment plan itself:
- What cell product is being used
- Whether the protocol uses cultured allogeneic MSCs or a more limited alternative
- How the treatment is matched to your diagnosis and goals
- How much physician evaluation and procedural oversight are included
- Whether pre-treatment review, diagnostics, and follow-up are part of the fee
- What quality documentation exists for testing, handling, and release
These details determine value far more than a headline price.
If a clinic in Mexico offers a well-characterized cell product, condition-specific planning, and physician-directed care for less than a narrower U.S. option, that is not a compromise. It may be the stronger medical decision.
Patients who want a broader framework for comparing pricing, inclusions, and treatment scope can review these stem cell therapy cost considerations.
The costly mistake patients make
The weakest way to compare clinics is to line up advertisements and choose the lowest number.
A better comparison is clinical. What is being administered? Why was that protocol chosen for your diagnosis? Who is responsible for the case if your response is incomplete? What follow-up is included?
Those are the questions that protect both your outcome and your budget. In regenerative medicine, paying less for the wrong treatment is more expensive than paying appropriately for the right one.
Your Patient Decision Checklist for Stem Cell Therapy
When patients feel overwhelmed, a checklist helps. Not because regenerative medicine is simple, but because the right questions quickly separate serious clinics from sales operations.
Clinic and lab credentials
Start with the foundation.
- Verify the clinic’s legal operating status. Ask what regulatory framework it works under and whether its laboratory relationships are documented.
- Ask where the cells are processed. If the clinic can’t tell you where the lab is or how it is licensed, stop there.
- Request quality documentation. A serious center should be comfortable discussing cell identity, viability, purity, and sterility procedures.
A clinic doesn’t need to overwhelm you with jargon. It does need to answer clearly.
The therapeutic protocol
In this area, many patients ask too few questions.
You want to know:
- What cells are being used?
Ask specifically whether the treatment uses cultured allogeneic MSCs and from what tissue source. - Why is this protocol recommended for my diagnosis?
Knee osteoarthritis, rotator cuff injury, autoimmune disease, and neuropathy are not interchangeable. - How will the treatment be delivered?
Joint injection, local injection, IV support, or a combination may all have different purposes. - What outcomes are realistic?
Good doctors discuss improvement in pain, function, recovery, or inflammation support. They don’t promise miracles.
The physician team
This part should never be skipped.
Look for:
- Named physicians who are visible and credentialed
- Direct access to the clinician making recommendations
- Experience with your condition, not just regenerative medicine in general
- A clear procedural plan for who performs the treatment
If you can’t identify the physician responsible for your care before you travel, you’re not choosing a medical program. You’re choosing a marketing funnel.
Evaluation and follow-up
Treatment quality starts before the procedure and continues after it.
A reliable clinic should explain:
- How your candidacy is assessed
- What prior imaging or records are needed
- What you should expect after treatment
- How follow-up communication works once you return home
- When further rehabilitation or monitoring is recommended
Some patients need a one-time orthopedic intervention. Others need a broader plan with physical medicine, movement support, or longitudinal monitoring.
Red-light and green-light signals
Here’s a practical screen.
Green lights
- Clear answers
- Transparent cell information
- Physician-led evaluation
- Realistic expectations
- Defined follow-up
Red lights
- Pressure to book quickly
- Guarantee language
- No lab transparency
- No individualized planning
- No discussion of candidacy limits
Patients don’t need to become stem cell scientists. They do need to become careful buyers of complex medical care.
Frequently Asked Questions About Treatment in Mexico
Is it legal for a U.S. or Canadian patient to travel to Mexico for stem cell therapy?
Yes. Patients commonly travel internationally for medical care, including regenerative care, when treatment is legally offered in the destination country. What matters is that the clinic is operating under the appropriate framework and that you understand the treatment, the clinic’s standards, and the follow-up plan.
Does treatment in Mexico mean lower standards?
Not necessarily.
Standards depend on the clinic, the physicians, and the laboratory systems behind the therapy. Some clinics in Mexico operate at a very high level. Others do not. The same broad principle applies everywhere. Patients should judge the program, not the marketing narrative around the country.
How are allogeneic stem cells sourced?
Clinics should explain this directly. Ethical sourcing, donor screening, tissue processing, and product release standards should all be part of the conversation. If a clinic avoids specifics, that’s a reason to pause.
What does the travel process usually look like?
In a well-run clinic, travel should feel structured. Patients usually complete record review and candidacy screening before arrival. The in-person portion may include consultation, exam, treatment, and recovery guidance. Better clinics also help coordinate logistics so the medical side doesn’t feel rushed.
What happens after I return home?
Aftercare should be discussed before treatment, not after it.
Patients should know who to contact with questions, what recovery milestones to expect, when activity should be modified, and whether additional care such as rehabilitation, reassessment, or supportive therapies may help. Good follow-up reduces uncertainty and improves decision quality even before the procedure takes place.
Is Mexico the right choice for everyone?
No.
Some patients are better served by conservative care, surgery, rehabilitation, or a different medical workup. Some should stay closer to home for continuity reasons. Others are excellent candidates for treatment in Mexico if they choose a physician-led clinic with strong lab standards and transparent protocols.
The best decision is rarely the most emotional one. It’s the one built on documentation, diagnosis, and realistic expectations.
If you’re considering regenerative care and want a physician-led assessment of whether treatment in Mexico is appropriate for your condition, Longevity Medical Institute offers advanced, personalized guidance in a premium clinical setting designed for U.S. and Canadian patients seeking clarity, quality, and continuity of care.
Published on April 14, 2026
Short Disclaimer
This information is for educational purposes only and is not medical advice. It does not replace an evaluation by a qualified healthcare professional. For personalized guidance, please schedule a consultation.